P0515
Phospholipase D from Arachis hypogaea (peanut)
Type II, lyophilized powder, ≥60 units/mg protein
Synonym(s):
Lecithinase D, Phosphatidylcholine phosphatidohydrolase
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Recommended Products
biological source
Arachis hypogaea
Quality Level
type
Type II
form
lyophilized powder
specific activity
≥60 units/mg protein
composition
Protein, ~30%
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
General description
Phospholipase D is a phospholipid hydrolyzing enzyme and an important component of receptor-mediated signal transduction responses and regulated secretion.
Phospholipase D (PLD) is abundantly found in the eukaryotes and prokaryotes. It is present in all mammalian cells. In mammals, PLD is initiated by various hormones, neurotransmitters, growth factors and cytokines.
Application
Phospholipase D from Arachis hypogaea (peanut) has been used in the preparation of lipase enzyme solution. It has also been used to determine its influence on human neutrophil respiratory function.
Research has shown ADP-ribosylation factor regulation of phospholipase D is important in the release of nascent secretory vesicles from the trans-Golgi network. It has also been used in a study to investigate stimulation of Na+-Ca2+ exchange activity in canine cardiac sarcolemmal vesicals.
Biochem/physiol Actions
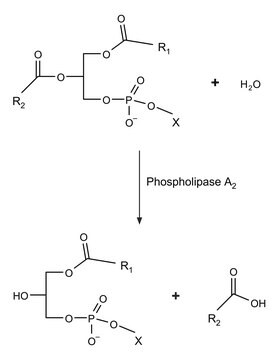
Hydrolyzes the phosphate bonds of phospholipids and sphingomyelin to give the corresponding phosphatidic acid.
Phospholipase D is involved in conferring drought susceptibility in peanuts, which increases the risk of aflatoxin contamination.
Phospholipase D (PLD) modulates cell growth, secretion and the actin cytoskeleton.
Unit Definition
One unit will liberate 1.0 μmol of choline from L-α-phosphatidylcholine (egg yolk) per hr at pH 5.6 at 30 °C.
Physical form
Partially purified, lyophilized powder containing buffer salts
Analysis Note
Protein determined using biuret, unless otherwise indicated.
inhibitor
Product No.
Description
Pricing
signalword
Danger
hcodes
pcodes
Hazard Classifications
Resp. Sens. 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Expression and characterization of rat brain phospholipase D
Xie Z, et al.
Methods in Enzymology, 345, 255-264 (2002)
Human neutrophil peptide receptors: mobilization mediated by phospholipase C.
Nelson R D, et al.
The American Journal of Pathology, 107(2), 202-202 (1982)
Giuseppe Dionisio et al.
Plant physiology, 156(3), 1087-1100 (2011-01-12)
Barley (Hordeum vulgare) and wheat (Triticum aestivum) possess significant phytase activity in the mature grains. Maize (Zea mays) and rice (Oryza sativa) possess little or virtually no preformed phytase activity in the mature grain and depend fully on de novo
K D Philipson et al.
The Journal of biological chemistry, 259(1), 16-19 (1984-01-10)
Treatment of canine cardiac sarcolemmal vesicles with phospholipase D resulted in a large stimulation (up to 400%) of Na+-Ca2+ exchange activity. The phospholipase D treatment decreased the apparent Km (Ca2+) for the initial rate of Nai+-dependent Ca2+ uptake from 18.2
B Z Guo et al.
Planta, 223(3), 512-520 (2005-10-04)
Preharvest aflatoxin contamination has been identified by the peanut industry as a serious issue in food safety and human health because of the carcinogenic toxicity. Drought stress is the most important environmental factor exacerbating Aspergillus infection and aflatoxin contamination in
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service