おすすめの製品
グレード
pharmaceutical primary standard
APIファミリー
pyrazinamide
メーカー/製品名
EDQM
mp
189-191 °C (lit.)
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
保管温度
2-8°C
SMILES記法
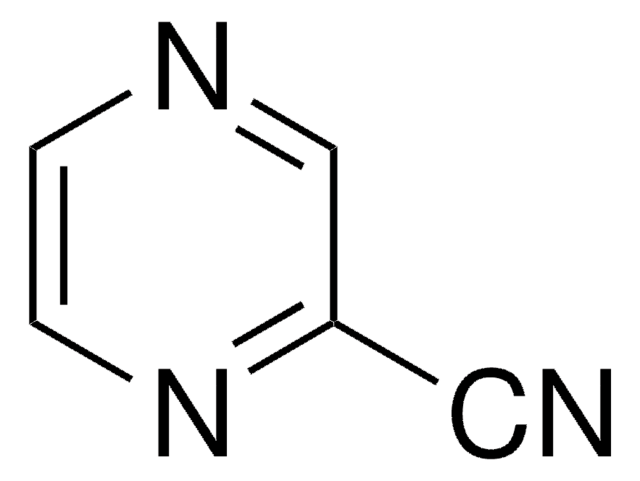
NC(=O)c1cnccn1
InChI
1S/C5H5N3O/c6-5(9)4-3-7-1-2-8-4/h1-3H,(H2,6,9)
InChI Key
IPEHBUMCGVEMRF-UHFFFAOYSA-N
類似した製品をお探しですか? 訪問 製品比較ガイド
詳細
この製品は薬局方標準品です。発行元の薬局方により製造・供給されています。MSDSを含む製品情報などの詳しい情報は、発行元の薬局方のウェブサイトよりご確認ください。
アプリケーション
Pyrazinamide EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
包装
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
その他情報
Sales restrictions may apply.
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
最新バージョンのいずれかを選択してください:
この製品を見ている人はこちらもチェック
D A Mitchison et al.
Tuberculosis (Edinburgh, Scotland), 90(3), 177-181 (2010-04-13)
While we wait for improved new anti-tuberculosis drugs, the main aim for improving current treatment should be to optimize the use of the two current drugs, rifampicin and the pro-drug pyrazinamide, which are responsible to a similar extent for the
X Gonzalo et al.
The Journal of antimicrobial chemotherapy, 69(11), 3001-3005 (2014-06-26)
Pyrazinamide is a key first-line tuberculosis drug. Reliable drug susceptibility testing (DST) data are of clinical importance, but in vitro testing is challenging since the activity of pyrazinamide is pH sensitive. The BACTEC MGIT 960 is considered the principal reference
Y Zhang et al.
The international journal of tuberculosis and lung disease : the official journal of the International Union against Tuberculosis and Lung Disease, 7(1), 6-21 (2003-04-19)
Pyrazinamide (PZA) is an important sterilising tuberculosis drug that helps to shorten the duration of current chemotherapy regimens for tuberculosis. When first discovered, it had activity in murine tuberculosis but no apparent in vitro activity, and its subsequent use in
L Gwaza et al.
Clinical pharmacology and therapeutics, 96(5), 580-588 (2014-07-06)
Approval of generic medicines is based on bioequivalence with the innovator product, but it is not unusual for generics to be interchanged with each other. This study investigated the differences in bioavailability between World Health Organization-prequalified antituberculosis generics by means
Tawanda Gumbo et al.
The Journal of antimicrobial chemotherapy, 69(9), 2420-2425 (2014-05-14)
To identify the pyrazinamide MIC above which standard combination therapy fails. MICs of pyrazinamide were determined for Mycobacterium tuberculosis isolates, cultured from 58 patients in a previous randomized clinical trial in Cape Town, South Africa. The MICs were determined using
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)