모든 사진(1)
About This Item
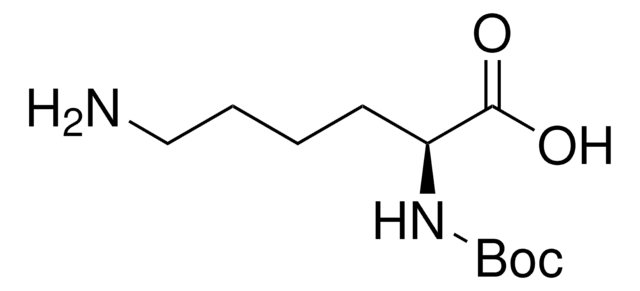
Linear Formula:
(CH3)3COCONH(CH2)4CH(NH2)COOH
CAS Number:
Molecular Weight:
246.30
Beilstein:
2417626
MDL number:
UNSPSC 코드:
12352209
PubChem Substance ID:
NACRES:
NA.22
추천 제품
product name
H-Lys(Boc)-OH, ≥95%
분석
≥95%
형태
powder
광학 활성
[α]20/D +18°, c = 1 in acetic acid
반응 적합성
reaction type: solution phase peptide synthesis
mp
250 °C (dec.) (lit.)
응용 분야
peptide synthesis
저장 온도
2-8°C
SMILES string
CC(C)(C)OC(=O)NCCCC[C@H](N)C(O)=O
InChI
1S/C11H22N2O4/c1-11(2,3)17-10(16)13-7-5-4-6-8(12)9(14)15/h8H,4-7,12H2,1-3H3,(H,13,16)(H,14,15)/t8-/m0/s1
InChI key
VVQIIIAZJXTLRE-QMMMGPOBSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
H-Lys(Boc)-OH also known as Nε-Boc-L-lysine, is commonly used in solution phase peptide synthesis.
애플리케이션
H-Lys(Boc)-OH can be used to prepare pentafluorophenyl esters which further used to synthesize β-peptides.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
Solution phase synthesis of beta-peptides using micro reactors
P Watts
Tetrahedron, 58, 5427-5439 (2002)
Maria Moccia et al.
International journal of pharmaceutics, 397(1-2), 179-183 (2010-07-06)
A novel nucleic acid compaction device based on a positively-charged alpha,epsilon-poly-l-lysine was realized for the first time. The polycationic peptide was obtained by assembling Fmoc and Boc orthogonally protected l-lysine monomers by solid phase synthesis. The route to the novel
April Case et al.
Analytical biochemistry, 338(2), 237-244 (2005-03-05)
Tissue transglutaminase (TGase) is a Ca(2+)-dependent enzyme that catalyzes cross-linking of intracellular proteins through a mechanism that involves isopeptide bond formation between Gln and Lys residues. In addition to its transamidation activity, TGase can bind guanosine 5'-triphosphate (GTP) and does
Ognyan K Argirov et al.
Biochimica et biophysica acta, 1620(1-3), 235-244 (2003-02-22)
Proteins are subject of posttranslational modification by sugars and their degradation products in vivo. The process is often referred as glycation. L-Dehydroascorbic acid (DHA), an oxidation product of L-ascorbic acid (vitamin C), is known as a potent glycation agent. A
K Itakura et al.
Chemical research in toxicology, 14(5), 473-475 (2001-05-23)
It has been suggested that protein modifications by malondialdehyde (MDA), a major product of lipid peroxidation, contribute to the fluorescence formation of lipofuscin. Although early studies proposed an aminoenimine structure (RNHCH=CHCH=NR) formed from MDA and the epsilon-amino groups of the
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.