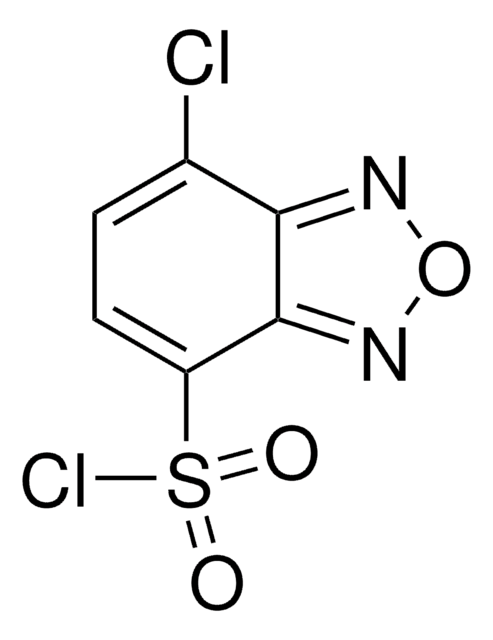
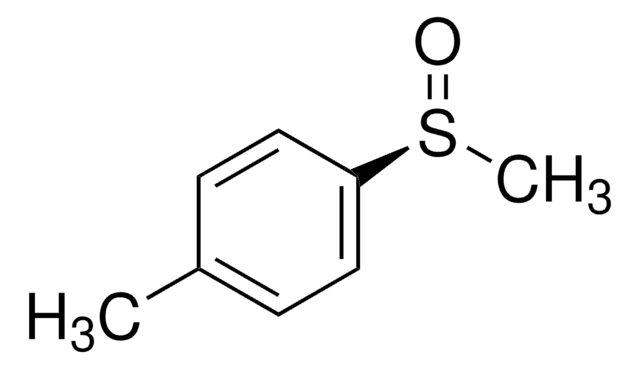
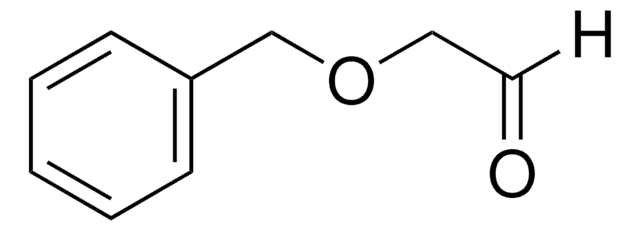
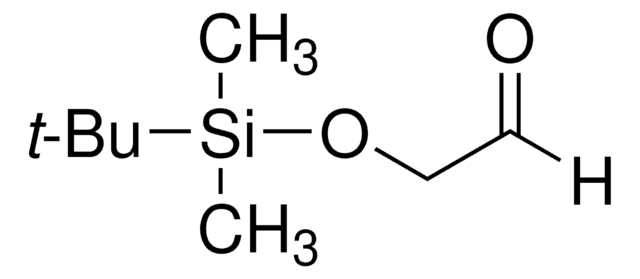
497401
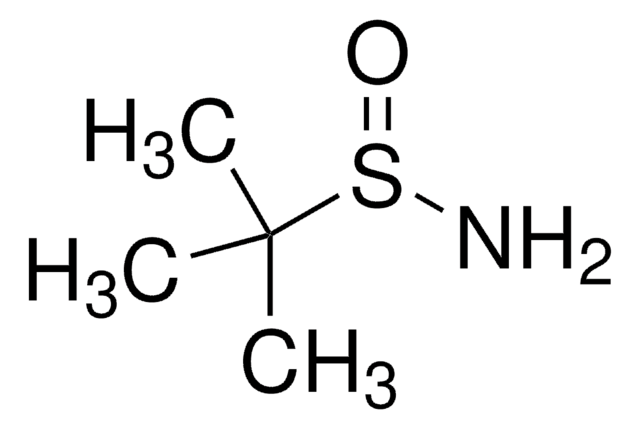
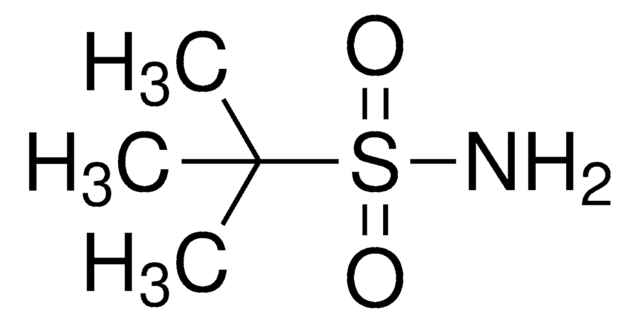
(R)-(+)-2-Methyl-2-propanesulfinamide
98%
동의어(들):
(R)-2-methyl-2-propanesulfinamide, (R)-2-methylpropane-2-sulfinamide, (R)-tert-butanesulfinamide, (R)-tert-butylsulfinamide
About This Item
추천 제품
Quality Level
분석
98%
광학 활성
[α]20/D +4°, c = 1.0242 in chloroform stab. with amylenes
mp
103-107 °C (lit.)
저장 온도
2-8°C
SMILES string
CC(C)(C)S(N)=O
InChI
1S/C4H11NOS/c1-4(2,3)7(5)6/h5H2,1-3H3/t7-/m1/s1
InChI key
CESUXLKAADQNTB-SSDOTTSWSA-N
일반 설명
애플리케이션
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
이미 열람한 고객
문서
Ellman's sulfinamide is available in both enantiomeric and racemic forms for your research. This versatile and useful auxiliary has found extensive use both in academics and industry.
관련 콘텐츠
Ellman group developed electron-rich phosphine ligands for C-H functionalization and tert-Butanesulfinamide for asymmetric amine synthesis.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.



![(R)-N-[(1R,2R)-2-(3-(3,5-Bis(trifluoromethyl)phenyl)ureido)cyclohexyl]-tert-butyl-sulfinamide 96%](/deepweb/assets/sigmaaldrich/product/structures/389/070/18847164-c6a7-4b4e-abcb-2dbc22493a2d/640/18847164-c6a7-4b4e-abcb-2dbc22493a2d.png)