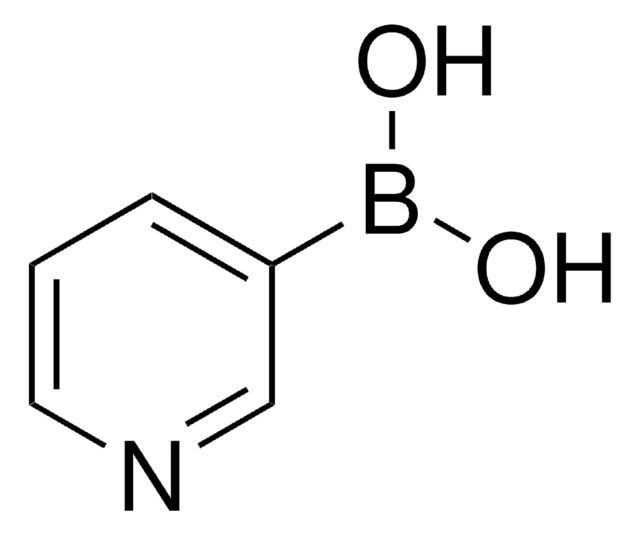
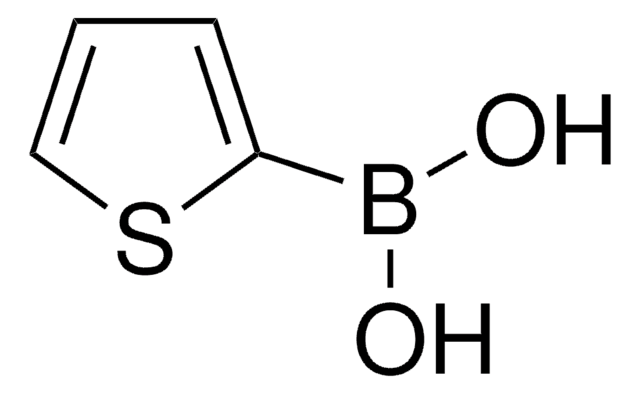
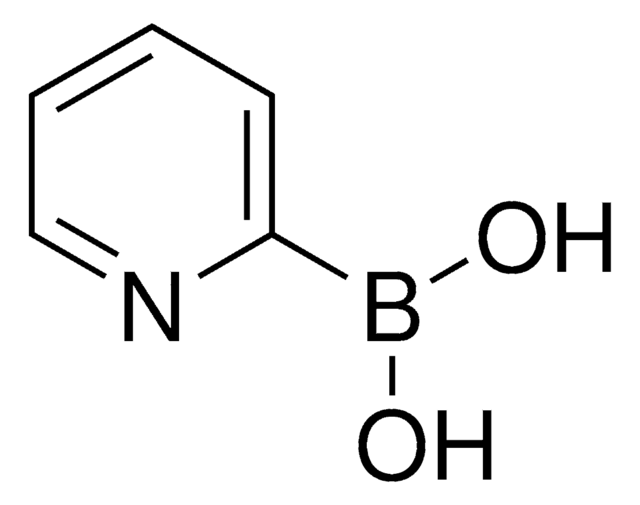
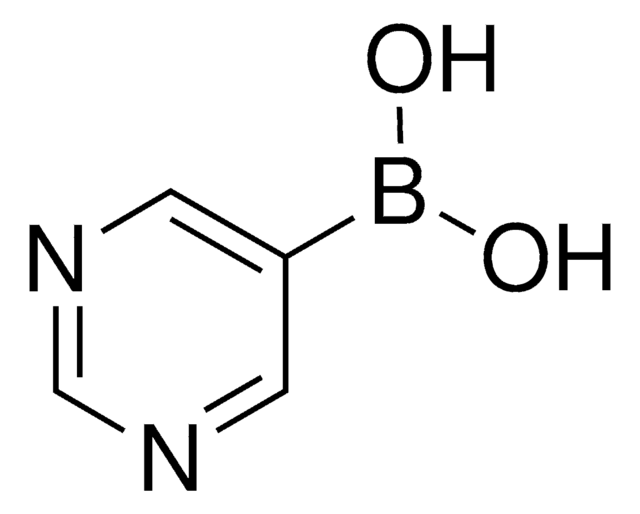
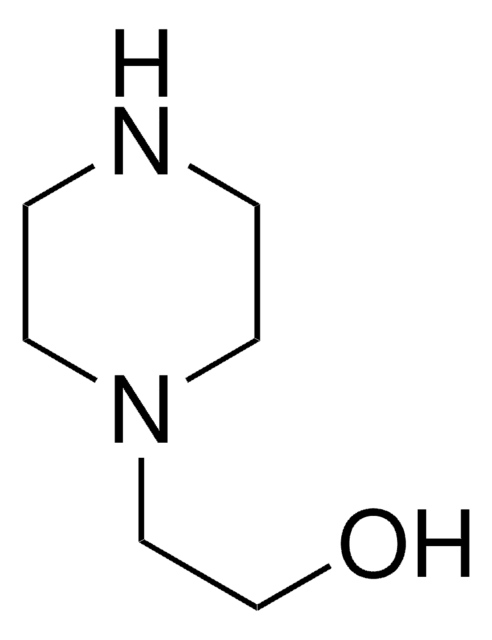
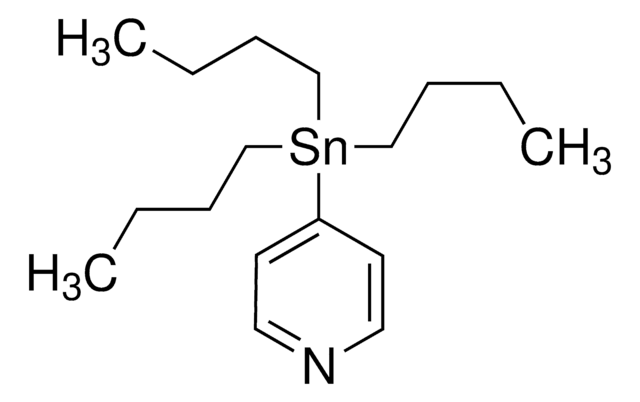
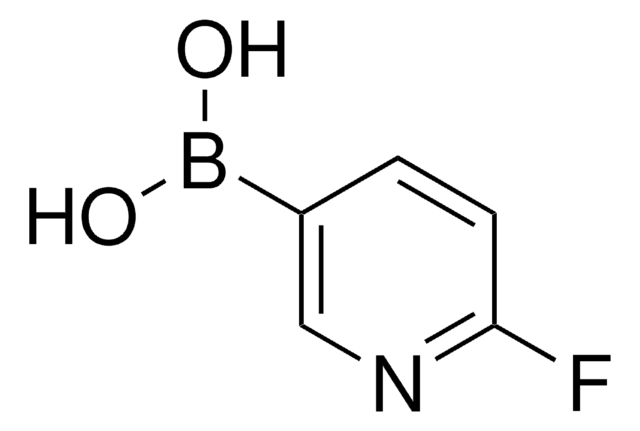
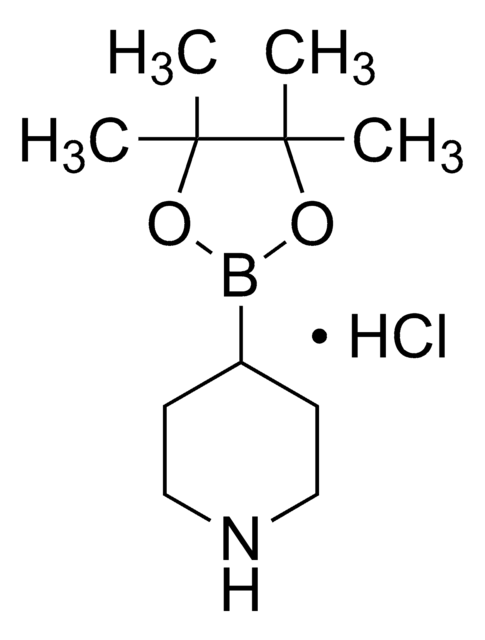
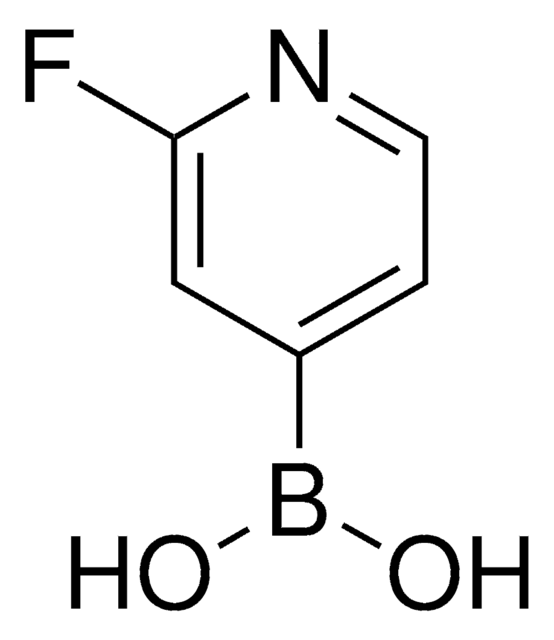
추천 제품
Quality Level
분석
90%
양식
solid
mp
>300 °C (lit.)
저장 온도
−20°C
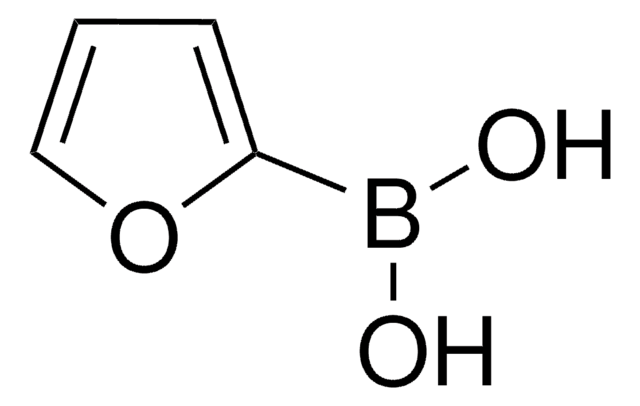
SMILES string
OB(O)c1ccncc1
InChI
1S/C5H6BNO2/c8-6(9)5-1-3-7-4-2-5/h1-4,8-9H
InChI key
QLULGIRFKAWHOJ-UHFFFAOYSA-N
일반 설명
4-Pyridinylboronic acid is commonly used as a reagent in cross-coupling reactions such as Suzuki-Miyaura cross-coupling.
애플리케이션
Reagent used for
Reagent used in Preparation of
- Palladium-catalyzed Suzuki-Miyaura coupling reactions
- Ligand-free palladium-catalyzed Suzuki coupling reaction under microwave irradation
Reagent used in Preparation of
- HIV-1 protease inhibitors
- Potential cancer threapeutics, such as PDK1 and protein kinase CK2 inhibitors
기타 정보
Contains varying amounts of anhydride
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
이미 열람한 고객
Sarah J Vella et al.
Beilstein journal of organic chemistry, 14, 1908-1916 (2018-08-17)
A two-station [2]catenane containing a large macrocycle with two different recognition sites, one bis(pyridinium)ethane and one benzylanilinium, as well as a smaller DB24C8 ring was synthesized and characterized. 1H NMR spectroscopy showed that the DB24C8 ring can shuttle between the
Jorge Cruz-Huerta et al.
Chemical communications (Cambridge, England), 48(35), 4241-4243 (2012-03-23)
The combination of two heteroaromatic boronic acids with pentaerythritol gave self-complementary tectons which were suitable for the generation of 2D and 3D molecular networks.
One-pot approach to N-quinolyl 3?/4?-biaryl carboxamides by microwave-assisted Suzuki--Miyaura coupling and N-boc deprotection
ZY Huang, et al.
The Journal of Organic Chemistry, 81, 9647-9657 (2016)
Jin-Tao Yu et al.
Organic & biomolecular chemistry, 10(7), 1359-1364 (2011-12-20)
An efficient palladium-catalyzed Suzuki-Miyaura coupling method involving the reaction between CTV-Br(3) and a variety of aryl and heteroaryl boronic acids in the presence of indolyl phosphane ligands has been developed. This reaction procedure provided a series of C(3)-symmetric aryl-extended rigid
Sumin Lee et al.
Organic letters, 14(9), 2238-2241 (2012-04-28)
The kinetic process of key intermediates involved in the electrochemical ring opening of photochromic dithienylcyclopentenes (DTEs) has been observed for the first time, where the electronic nature of the DTEs is an important factor that determines the rate-determining step in
문서
Suzuki-Miyaura cross-coupling reaction is extensively used in organic chemistry, polymer science, and pharmaceutical industries.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.