727970
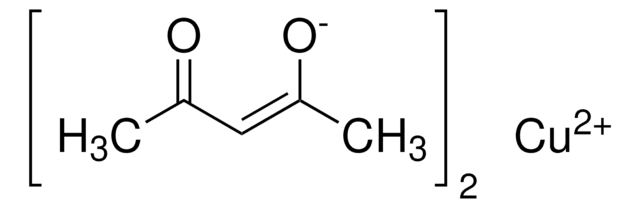
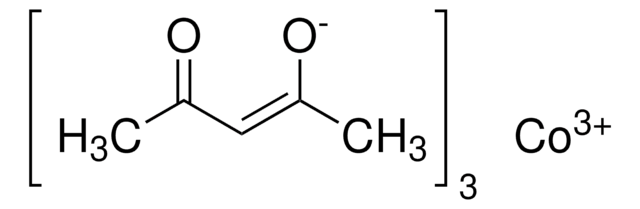
Cobalt(II) acetylacetonate
≥99.0% (KT)
동의어(들):
2,4-Pentanedione cobalt(II) derivative, Bis(2,4-pentanedionato)cobalt, Co(acac)2, Cobaltous acetylacetonate
About This Item
추천 제품
분석
≥99.0% (KT)
형태
powder
반응 적합성
core: cobalt
reagent type: catalyst
입자 크기
≤20 mesh
mp
165-170 °C (lit.)
음이온 미량물
chloride, bromide (as Cl)-: ≤500 mg/kg
양이온 미량물
Ca: ≤50 mg/kg
Cd: ≤50 mg
Co: ≤50 mg/kg
Cu: ≤50 mg/kg
Fe: ≤50 mg/kg
K: ≤100 mg/kg
Na: ≤100 mg/kg
Ni: ≤50 mg/kg
Pb: ≤50 mg/kg
Zn: ≤50 mg/kg
SMILES string
CC(=O)\C=C(\C)O[Co]O\C(C)=C/C(C)=O
InChI
1S/2C5H8O2.Co/c2*1-4(6)3-5(2)7;/h2*3,6H,1-2H3;/q;;+2/p-2/b2*4-3-;
InChI key
UTYYEGLZLFAFDI-FDGPNNRMSA-L
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
애플리케이션
Cobalt(II) acetylacetonate can be used as a catalyst for the dehydration reaction of aldoximes to synthesize nitriles.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1B Inhalation - Eye Dam. 1 - Repr. 1B - Resp. Sens. 1 - Skin Sens. 1
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.



![Thieno[2,3-b]thiophene 95%](/deepweb/assets/sigmaaldrich/product/structures/338/511/a37343cf-31cb-4a34-b903-23b3dc8e64ab/640/a37343cf-31cb-4a34-b903-23b3dc8e64ab.png)