추천 제품
Grade
certified reference material
Quality Level
형태
liquid
특징
(Snap-N-Spike®)
포장
ampule of 1 mL
제조업체/상표
Cerilliant®
농도
1.0 mg/mL in acetonitrile
기술
gas chromatography (GC): suitable
liquid chromatography (LC): suitable
응용 분야
clinical testing
형식
single component solution
배송 상태
dry ice
저장 온도
−70°C
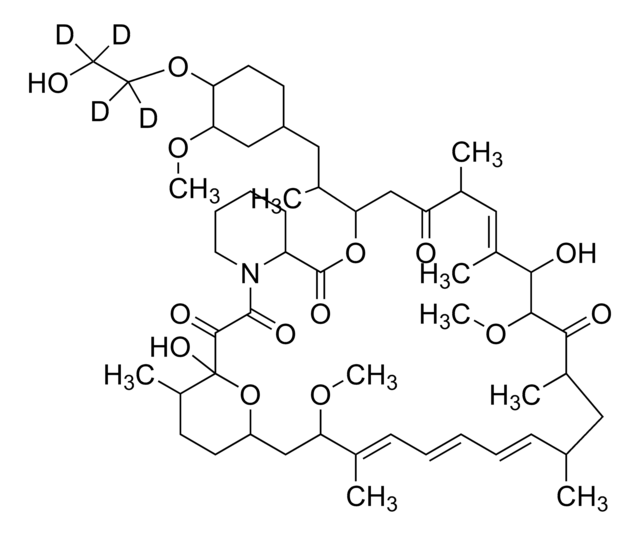
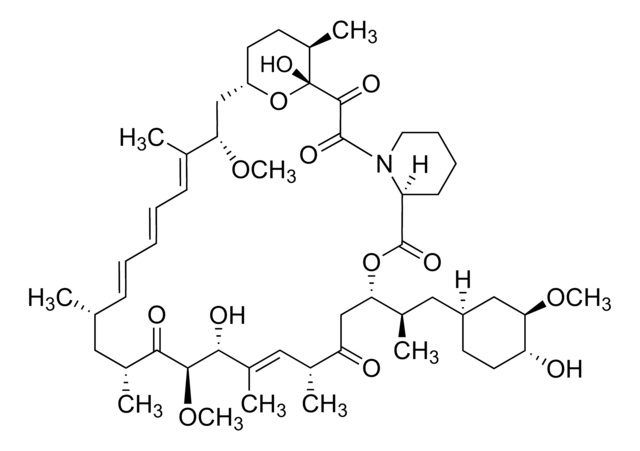
InChI
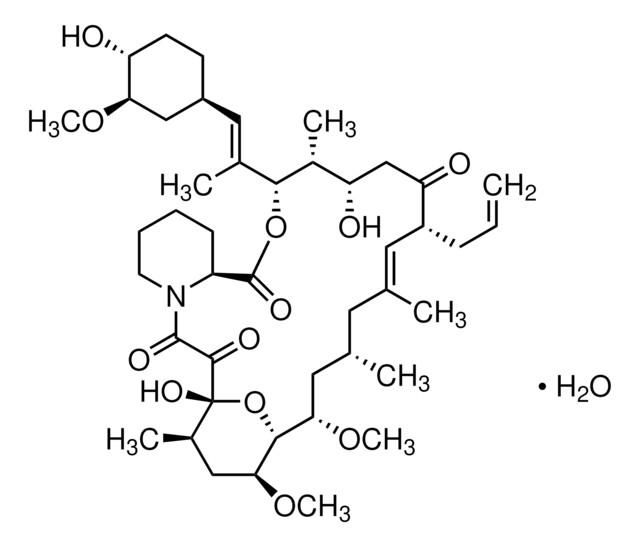
1S/C53H83NO14/c1-32-16-12-11-13-17-33(2)44(63-8)30-40-21-19-38(7)53(62,68-40)50(59)51(60)54-23-15-14-18-41(54)52(61)67-45(35(4)28-39-20-22-43(66-25-24-55)46(29-39)64-9)31-42(56)34(3)27-37(6)48(58)49(65-10)47(57)36(5)26-32/h11-13,16-17,27,32,34-36,38-41,43-46,48-49,55,58,62H,14-15,18-26,28-31H2,1-10H3/b13-11+,16-12+,33-17+,37-27+/t32-,34-,35-,36-,38-,39+,40+,41+,43-,44+,45+,46-,48-,49+,53-/m1/s1
InChI key
HKVAMNSJSFKALM-GKUWKFKPSA-N
유전자 정보
human ... FKBP1A(2280)
일반 설명
애플리케이션
- Everolimus as a therapy for hepatoblastoma: Research demonstrates that Everolimus can induce autophagy-dependent ferroptosis in hepatoblastoma cells, highlighting its potential as a therapeutic agent in oncology research. This study provides insight into the mechanisms by which Everolimus can be utilized to target cancer cells through cell death pathways (Huang et al., 2024).
- Understanding oral mucosal injuries from mTOR inhibitors: A new hypothesis posits that oral mucosal injuries associated with mTOR inhibitors like Everolimus result from disruptions in cellular stress and apoptotic pathways. This study underscores the importance of understanding side effects in the context of targeted therapy for conditions such as cancers and immunosuppression (Sonis and Villa, 2023).
- Micellar formulation of Everolimus for neurological disorders: A stable micellar formulation of Everolimus (RAD001) has been developed for intracerebroventricular delivery, aimed at treating Alzheimer′s Disease and other neurological disorders. This formulation allows for direct brain administration, potentially enhancing the drug′s efficacy and safety profile (Gianessi et al., 2023).
- Pharmacokinetics in epilepsy treatment: The population pharmacokinetics of Everolimus were studied in patients with seizures associated with focal cortical dysplasia. This research aids in understanding the drug′s behavior in a specific neurological context, providing a foundation for dosing adjustments and therapeutic monitoring (Park et al., 2023).
법적 정보
관련 제품
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Flam. Liq. 2
Storage Class Code
3 - Flammable liquids
WGK
WGK 2
Flash Point (°F)
35.6 °F - closed cup
Flash Point (°C)
2.0 °C - closed cup
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.