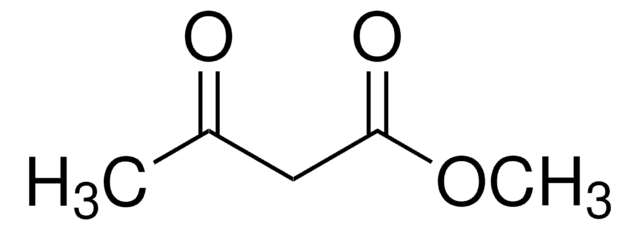
추천 제품
vapor pressure
0.26 hPa ( 20 °C)
Quality Level
분석
≥98.0% (GC)
형태
liquid
autoignition temp.
350 °C
효능
3980 mg/kg LD50, oral (Rat)
>5000 mg/kg LD50, skin (Rabbit)
expl. lim.
1.0-54 % (v/v)
pH
4.0 (20 °C, 110 g/L in H2O)
mp
-53.3 °C
전이 온도
flash point 73.5 °C
solubility
130.3 g/L
density
1.03 g/cm3 at 20 °C
저장 온도
2-30°C
InChI
1S/C6H10O3/c1-3-9-6(8)4-5(2)7/h3-4H2,1-2H3
InChI key
XYIBRDXRRQCHLP-UHFFFAOYSA-N
애플리케이션
Ethyl acetoacetate can be used as a reactant to synthesize:
It can be also utilized as a reactant in the transesterification and asymmetric hydrogenation reactions to produce valuable products.
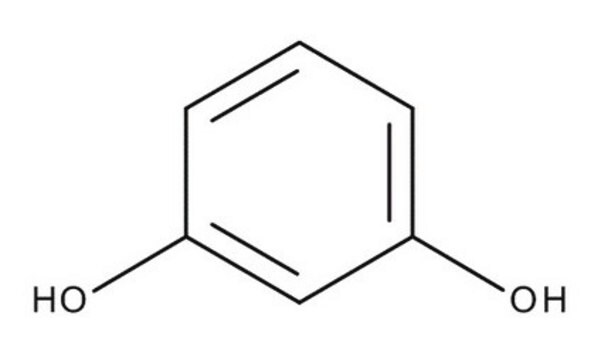
- 7-hydroxycoumarin derivatives via Pechmann condensation reaction with 1,3-dihydroxybenzene in the presence of acid catalysts.
- 2,6-disubstituted piperidine alkaloid, (−)-pinidinone via stereoselective α-aminoallylation followed by Grubbs′ olefin cross-metathesis reaction.
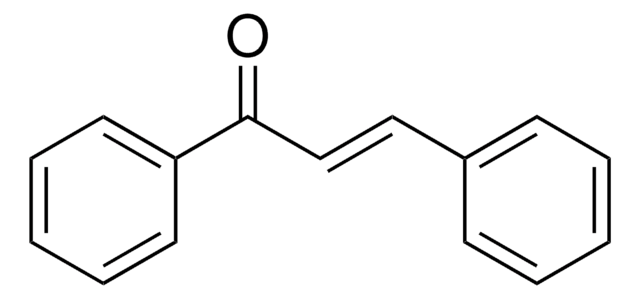
- Michael addition products via Michael addition reaction with chalcones and azachalcones in the presence of a base catalyst.
- α, β-unsaturated carbonyl compounds via Knoevenagel condensation reaction with glyceraldehyde acetonide in the presence of a base catalyst.
It can be also utilized as a reactant in the transesterification and asymmetric hydrogenation reactions to produce valuable products.
분석 메모
Assay (GC, area%): ≥ 98.0 % (a/a)
Density (d 20 °C/ 4 °C): 1.028 - 1.030
Identity (IR): passes test
Density (d 20 °C/ 4 °C): 1.028 - 1.030
Identity (IR): passes test
Storage Class Code
10 - Combustible liquids
WGK
WGK 1
Flash Point (°F)
164.3 °F - closed cup
Flash Point (°C)
73.5 °C - closed cup
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
An Expeditious Stereoselective Synthesis of (-)-Pinidinone from Ethyl Acetoacetate
Damodar K and Jun Jong-Gab
Bulletin of the Korean Chemical Society,, 37(4), 571-575 (2016)
Synthesis of 7-hydroxycoumarins catalysed by solid acid catalysts
Hoefnagel AJ, et al.
Journal of the Chemical Society. Chemical Communications, (2), 225-226 (1995)
Condensation of glyceraldehyde acetonide with ethyl acetoacetate over Mg, Al-mixed oxides derived from hydrotalcites
Veloso Claudia O, et al.
Microporous and Mesoporous Materials : The Official Journal of the International Zeolite Association, 107(1-2), 23-30 (2008)
Mechanochemical Michael reactions of chalcones and azachalcones with ethyl acetoacetate catalyzed by K2CO3 under solvent-free conditions
Zhang Ze, et al.
Chemistry Letters (Jpn), 33(2), 168-169 (2004)
Boric acid: an efficient and environmentally benign catalyst for transesterification of ethyl acetoacetate
Kondaiah GCM, et al.
Tetrahedron Letters, 49(1), 106-109 (2008)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.