모든 사진(1)
About This Item
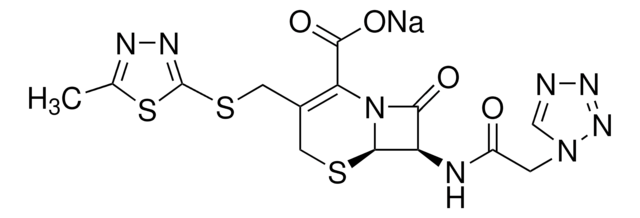
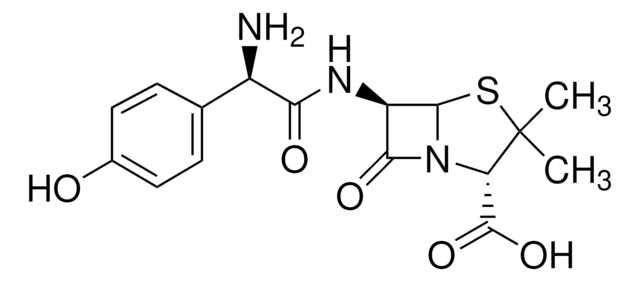
실험식(Hill 표기법):
C14H14N8O4S3
CAS Number:
Molecular Weight:
454.51
UNSPSC 코드:
41116107
NACRES:
NA.24
추천 제품
Grade
pharmaceutical primary standard
API family
cefazolin
제조업체/상표
EDQM
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-8°C
InChI
1S/C14H14N8O4S3/c1-6-17-18-14(29-6)28-4-7-3-27-12-9(11(24)22(12)10(7)13(25)26)16-8(23)2-21-5-15-19-20-21/h5,9,12H,2-4H2,1H3,(H,16,23)(H,25,26)/t9-,12-/m1/s1
InChI key
MLYYVTUWGNIJIB-BXKDBHETSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
애플리케이션
Cefazolin EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
포장
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
기타 정보
Sales restrictions may apply.
관련 제품
제품 번호
설명
가격
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Resp. Sens. 1 - Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
이미 열람한 고객
Angela Macaluso et al.
The journal of pain : official journal of the American Pain Society, 14(6), 604-612 (2013-06-04)
Repeated injections of the antibiotic ceftriaxone cause analgesia in rodents by upregulating the glutamate transporter, GLT-1. No evidence is available in humans. We studied the effect of a single intravenous administration of ceftriaxone in patients undergoing decompressive surgery of the
John D Turnidge
Clinical infectious diseases : an official publication of the Infectious Diseases Society of America, 52(7), 917-924 (2011-03-24)
Clinical and Laboratory Standards Institute breakpoints for cefazolin against Enterobacteriaceae that were published in January 2010 have been revised by the Subcommittee on Antimicrobial Susceptibility Testing, based on the examination of recent data about in vitro activity, pharmacokinetic-pharmacodynamic characteristics and
Mack W Savage et al.
American journal of obstetrics and gynecology, 209(2), 108-108 (2013-05-29)
To identify risk factors for and outcomes of surgical site infections and cellulitis after abdominal hysterectomies. We used logistic regression analysis to analyze data from a case-control study of 1104 patients undergoing abdominal hysterectomies at a university hospital between Jan.
T Osler et al.
Diseases of the colon and rectum, 29(2), 140-143 (1986-02-01)
The seventh case of probable cefazolin-induced pseudomembranous colitis is reported. Perforation of the colon necessitated sigmoid resection. The postoperative course was protracted, and illustrates the difficulty of managing advanced pseudomembranous colitis when the oral route of antibiotic administration is not
Nagaraju Konda et al.
Optometry and vision science : official publication of the American Academy of Optometry, 91(1), 47-53 (2013-11-12)
To investigate the utility of additional microbial analyses to detect the causative microorganism of microbial keratitis and to determine the strength of association between clinical variables. This retrospective study included 125 eyes of 123 people presenting with presumed infectious keratitis
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.