추천 제품
Quality Level
분석
≥98% (TLC)
양식
powder
광학 활성
[α]/D 123.00 to 135.00°, c = 9.00-11.00 mg/mL in methanol
기술
thin layer chromatography (TLC): suitable
색상
white to yellow cast
solubility
H2O: soluble
저장 온도
2-8°C
SMILES string
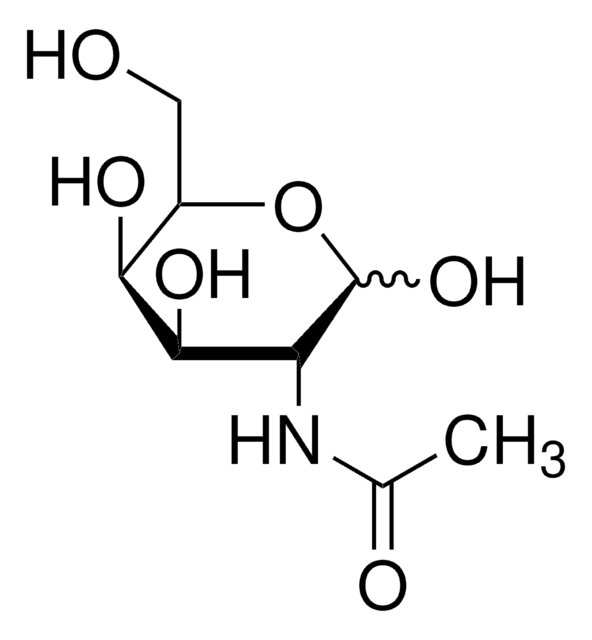
Nc1ccc(OC2OC(CO)C(O)C(O)C2O)cc1
InChI
1S/C12H17NO6/c13-6-1-3-7(4-2-6)18-12-11(17)10(16)9(15)8(5-14)19-12/h1-4,8-12,14-17H,5,13H2
InChI key
MIAKOEWBCMPCQR-UHFFFAOYSA-N
애플리케이션
4-Aminophenyl α-D-mannopyranoside is used to modify the surface of liposomes to increase uptake kinetics.
기타 정보
To gain a comprehensive understanding of our extensive range of Monosaccharides for your research, we encourage you to visit our Carbohydrates Category page.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
Lot/Batch Number
이미 열람한 고객
Sumio Chono et al.
Drug development and industrial pharmacy, 36(1), 102-107 (2009-08-07)
The effect of surface-mannose modification on aerosolized liposomal delivery to alveolar macrophages (AMs) was evaluated in vitro and in vivo. 4-Aminophenyl-α-D-mannopyranoside (Man) was used for surface-mannose modification, and mannosylated liposomes with various mannosylation rates (particle size: 1000 nm) were prepared.
Xue Ying et al.
Pharmacology, 87(1-2), 105-114 (2011-02-02)
To circumvent the problem of transporting anticancer drugs across the blood-brain barrier (BBB) to target brain tumors, we have previously developed dual-targeting daunorubicin liposomes modified with 4-aminophenyl-α-D-manno-pyranoside and transferrin molecules. The objective of the present study was to evaluate the
Xue Ying et al.
Journal of controlled release : official journal of the Controlled Release Society, 141(2), 183-192 (2009-10-06)
Chemotherapy for brain glioma has been of limited value due to the inability of transport of drug across the blood-brain barrier (BBB) and poor penetration of drug into the tumor. For overcoming these hurdles, the dual-targeting daunorubicin liposomes were developed
M Triggiani et al.
Journal of immunology (Baltimore, Md. : 1950), 164(9), 4908-4915 (2000-04-26)
Secretory phospholipases A2 (sPLA2s) are a group of extracellular enzymes that release fatty acids at the sn-2 position of phospholipids. Group IIA sPLA2 has been detected in inflammatory fluids, and its plasma level is increased in inflammatory diseases. To investigate
Sofie Tanghe et al.
Molecular reproduction and development, 67(2), 224-232 (2003-12-25)
Several steps in the process of mammalian fertilization are mediated by carbohydrates. This study investigated the role of the p-aminophenyl derivative of d-mannose (APMP) during bovine fertilization. Inseminating cumulus-oocyte complexes (COCs) in the presence of increasing APMP concentrations resulted in
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.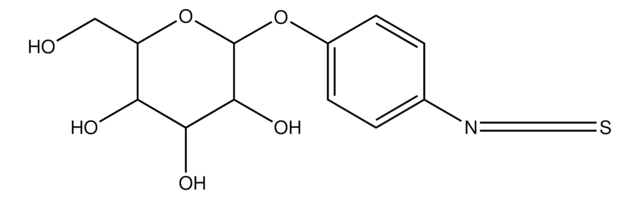







![(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethyl N-succinimidyl carbonate for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/969/022/d6776082-2f7a-47c7-bcd4-3830dac0fb7d/640/d6776082-2f7a-47c7-bcd4-3830dac0fb7d.png)
