129453
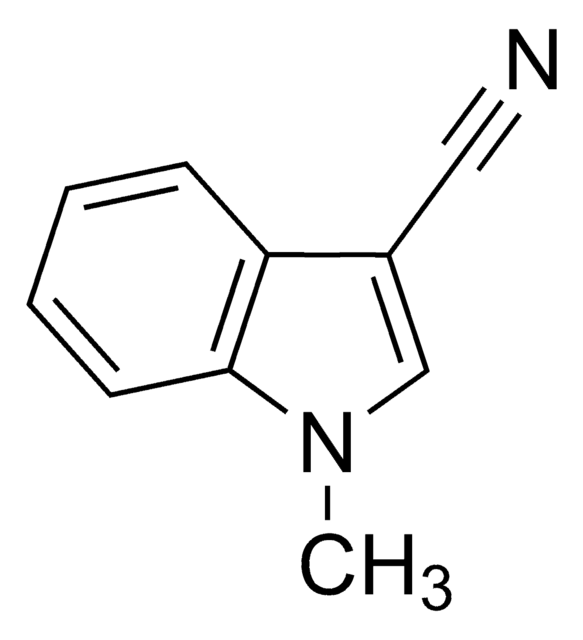
3-Indoleacetonitrile
98%
Synonym(s):
(3-Indolyl)acetonitrile, 3-(Cyanomethyl)indole, IAN, Indolylacetonitrile, NSC 523272
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C10H8N2
CAS Number:
Molecular Weight:
156.18
Beilstein/REAXYS Number:
125488
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
98%
form
solid
bp
157-160 °C/0.2 mmHg (lit.)
mp
33-36 °C (lit.)
functional group
nitrile
SMILES string
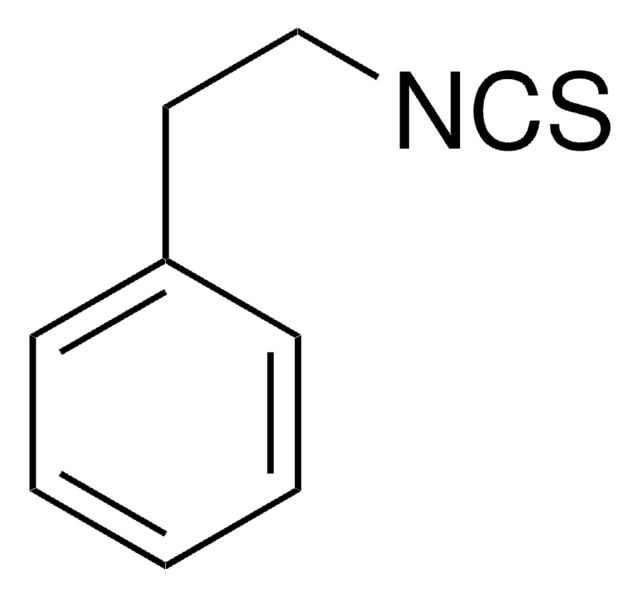
N#CCc1c[nH]c2ccccc12
InChI
1S/C10H8N2/c11-6-5-8-7-12-10-4-2-1-3-9(8)10/h1-4,7,12H,5H2
InChI key
DMCPFOBLJMLSNX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
3-Indoleacetonitrile (Indolylacetonitrile) is a light-induced auxin-inhibitory substance that is isolated from light-grown cabbage (Brassica olearea L.) shoots. It inhibits the biofilm formation of both E. coli O157:H7 and P. aeruginosa without affecting its growth.
Application
Reactant for preparation of:
- Tryptophan dioxygenase inhibitors pyridyl-ethenyl-indoles as potential anticancer immunomodulators
- Histone deacetylase inhibitors
- Potential kinase inhibitors
- Kv7/KCNQ potassium channel activators
- Kinesin-Specific MKLP-2 Inhibitor
- Pesticides
- Potential PET cancer imaging agents
- Agonists of the Farnesoid X Receptor (FXR) as atherosclerosis treatment
- Butyrylcholinesterase inhibitors
- Necroptosis inhibitors
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
233.6 °F - closed cup
flash_point_c
112 °C - closed cup
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Light-induced auxin-inhibiting substance from cabbage (Brassica oleacea L.) shoots.
Kosemura S, et al.
Tetrahedron Letters, 38(48), 8327-8330 (1997)
Alexandra Kutz et al.
The Plant journal : for cell and molecular biology, 30(1), 95-106 (2002-04-23)
Arabidopsis thaliana expresses four nitrilases, three of which (NIT1, NIT2 and NIT3) are able to convert indole-3-acetonitrile to indole-3-acetic acid (IAA), the plant growth hormone, while the isozyme NIT4 is a beta-cyano-l-alanine hydratase/nitrilase. NIT3 promoter activity is marginal in leaves
Pedro Robles et al.
Plant physiology, 152(3), 1357-1372 (2010-01-02)
To identify genes involved in vascular patterning in Arabidopsis (Arabidopsis thaliana), we screened for abnormal venation patterns in a large collection of leaf shape mutants isolated in our laboratory. The rotunda1-1 (ron1-1) mutant, initially isolated because of its rounded leaves
Nicole K Clay et al.
Science (New York, N.Y.), 323(5910), 95-101 (2008-12-20)
The perception of pathogen or microbe-associated molecular pattern molecules by plants triggers a basal defense response analogous to animal innate immunity and is defined partly by the deposition of the glucan polymer callose at the cell wall at the site
Yukako Komaki et al.
Journal of environmental sciences (China), 58, 231-238 (2017-08-05)
Disinfection by-products occur widely as the unintended effect of water disinfection and are associated with toxicity and adverse human health effects. Yet the molecular mechanisms of their toxicity are not well understood. To investigate the molecular basis of hyperploidy induction
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service