All Photos(1)
About This Item
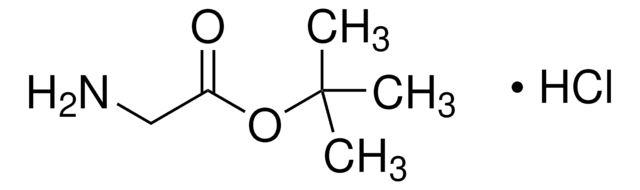
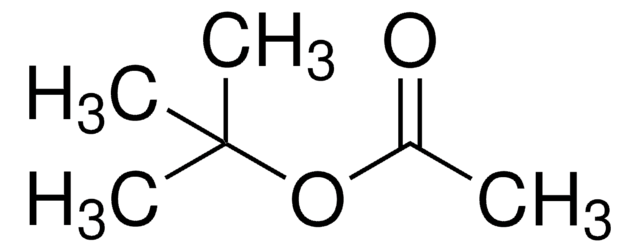
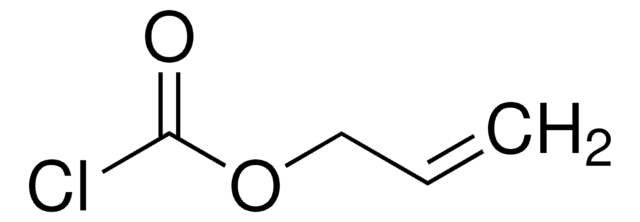
Linear Formula:
ClCH2COOC(CH3)3
CAS Number:
Molecular Weight:
150.60
Beilstein/REAXYS Number:
1753006
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
97%
refractive index
n20/D 1.423 (lit.)
bp
48-49 °C/11 mmHg (lit.)
density
1.053 g/mL at 25 °C (lit.)
functional group
chloro
ester
SMILES string
CC(C)(C)OC(=O)CCl
InChI
1S/C6H11ClO2/c1-6(2,3)9-5(8)4-7/h4H2,1-3H3
InChI key
KUYMVWXKHQSIAS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
tert-Butyl chloroacetate was used in the synthesis of:
- imidazol-1-yl-acetic acid hydrochloride
- cis-disubstituted aziridine ester via aza-Darzens reaction
- 1,10-diaza-18-crown-6 based sensors bearing a coumarin fluorophore
signalword
Danger
Hazard Classifications
Acute Tox. 3 Inhalation - Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Eye Irrit. 2 - Flam. Liq. 3 - Skin Irrit. 2
wgk_germany
WGK 1
flash_point_f
114.8 °F
flash_point_c
46 °C
ppe
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Santosh Kumar Singh et al.
Beilstein journal of organic chemistry, 4, 42-42 (2008-12-24)
A convenient and practical synthesis of imidazol-1-yl-acetic acid hydrochloride was achieved via N-alkylation of imidazole using tert-butyl chloroacetate followed by a non-aqueous ester cleavage of the resulting imidazol-1-yl-acetic acid tert-butyl ester in the presence of titanium tetrachloride. The synthesized imidazol-1-yl-acetic
Substituent dependent fluorescence response of diazacrown-based PET sensors.
Nagy K, et al.
Tetrahedron, 64(27), 6191-6195 (2008)
E Vedejs et al.
The Journal of organic chemistry, 65(18), 5498-5505 (2000-09-02)
Intermolecular alkylation of the aziridinyl oxazole 20 using PhSO(2)CH(2)CH(2)OTf is possible despite the presence of potentially nucleophilic aziridine nitrogen. The resulting oxazolium salt 22 reacts with BnNMe(3)(+)CN(-) to produce the azomethine ylide 24b via electrocyclic ring opening of an oxazoline
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service