292710
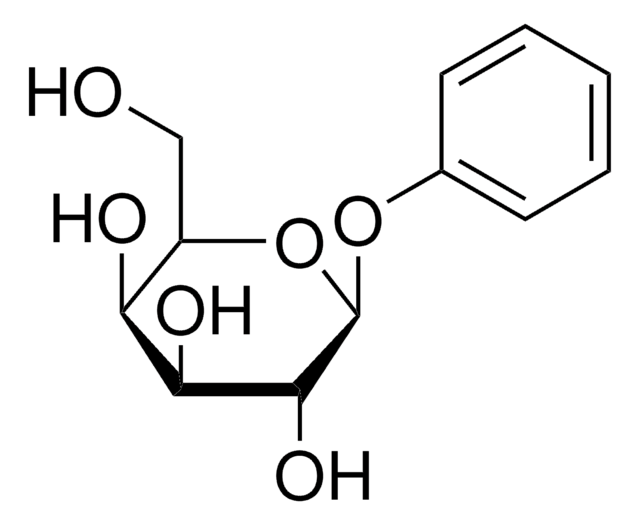
Phenyl β-D-glucopyranoside
≥95.0%
Synonym(s):
Phenyl beta-D-glucoside
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C12H16O6
CAS Number:
Molecular Weight:
256.25
Beilstein/REAXYS Number:
87517
EC Number:
MDL number:
UNSPSC Code:
12352201
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
≥95.0%
form
powder
optical activity
[α]25/D −70°, c = 1 in H2O
mp
176-178 °C (lit.)
SMILES string
OC[C@H]1O[C@@H](Oc2ccccc2)[C@H](O)[C@@H](O)[C@@H]1O
InChI
1S/C12H16O6/c13-6-8-9(14)10(15)11(16)12(18-8)17-7-4-2-1-3-5-7/h1-5,8-16H,6H2/t8-,9-,10+,11-,12-/m1/s1
InChI key
NEZJDVYDSZTRFS-RMPHRYRLSA-N
Application
Phenyl β-D-glucopyranoside can be used:
- As a starting material for the synthesis of various derivatives of β-D-glucopyranosides with potential application as anti-HIV agents.
- As a model for glycosides in the gas phase for their spectroscopic investigation.
- As an internal standard in GC and GC-MS quantitative analyses.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Synthesis of peptidomimetics based on iminosugar and ?-d-glucopyranoside scaffolds and inhibiton of HIV-protease.
Chery F, et al.
Tetrahedron, 60(31), 6597-6608 (2004)
Anke Reinders et al.
Plant, cell & environment, 29(10), 1871-1880 (2006-08-26)
Plant sucrose transporters (SUTs) are members of the glycoside-pentoside-hexuronide (GPH) cation symporter family (TC2.A.2) that is part of the major facilitator superfamily (MFS). All plant SUTs characterized to date function as proton-coupled symporters and catalyze the cellular uptake of sucrose.
Sugars in the gas phase: the spectroscopy and structure of jet-cooled phenyl ?-D-glucopyranoside.
Talbot FO and Simons JP
Physical Chemistry Chemical Physics, 4(15), 3562-3565 (2002)
4-(Arylamino)phenyl alpha-D-glucopyranosides as potential anti-HIV agents.
J C Briggs et al.
Carbohydrate research, 282(2), 293-298 (1996-03-18)
V T Edwards et al.
Xenobiotica; the fate of foreign compounds in biological systems, 16(9), 801-807 (1986-09-01)
The metabolic fates of 14C-phenol and its model plant conjugates 14C-phenyl glucoside and 14C-phenyl 6-O-malonyl-glucoside have been compared following equimolar oral dosing to rats (1.2 mg phenol/kg). Rapid excretion of radioactivity in the urine (at least 80% within 24 h)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service