All Photos(2)
About This Item
Empirical Formula (Hill Notation):
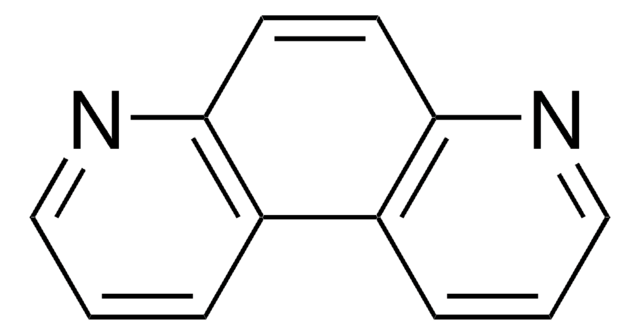
C12H8N2
CAS Number:
Molecular Weight:
180.21
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
99%
form
solid
mp
79-81 °C (lit.)
SMILES string
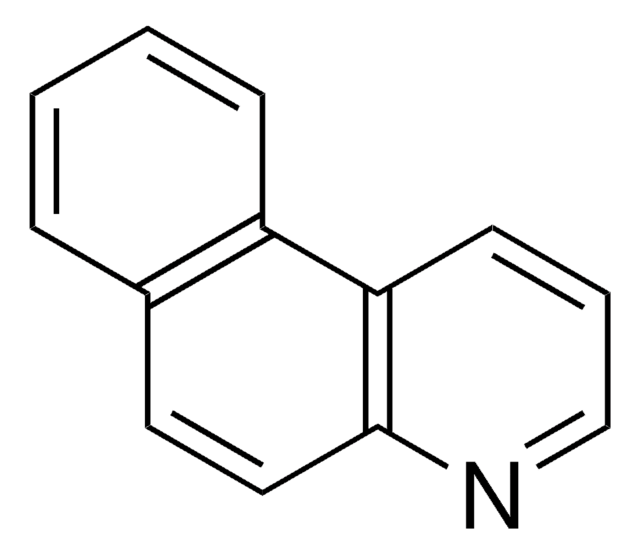
c1cnc2c(c1)ccc3ncccc23
InChI
1S/C12H8N2/c1-3-9-5-6-11-10(4-2-7-13-11)12(9)14-8-1/h1-8H
InChI key
OZKOMUDCMCEDTM-UHFFFAOYSA-N
General description
1,7-Phenanthroline has inhibitory effect on germination and growth of plants. 1,7-Phenanthroline is an aza-analog of phenanthrene, was found to be mutagenic in the Ames test using Salmonella typhimurium TA100 in the presence of a rat liver S9 fraction. Supramolecular assemblies of 1,2,4,5-benzenetetracarboxylic acid with 1,7-phenanthroline (aza donor molecule) were investigated.
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Kapildev K Arora et al.
The Journal of organic chemistry, 68(24), 9177-9185 (2003-11-25)
Supramolecular assemblies of 1,2,4,5-benzenetetracarboxylic acid, 1, with aza donor molecules such as 1,10-phenanthroline, 2, 1,7-phenanthroline, 3, phenazine, 4, 4-(N,N-dimethylamino)pyridine, 5, 1,2-bis(4-pyridyl)ethene, 6, and 1,2-bis(4-pyridyl)ethane, 7, have been synthesized and characterized by single-crystal X-ray diffraction methods. All the complexes crystallize in
Wei Xiao et al.
Environmental science & technology, 52(1), 114-123 (2017-12-06)
Iron (oxyhydr)oxides are widespread in natural and engineered systems, potent adsorbents of contaminants and a source of energy for iron-reducing bacteria. Microbial reduction of iron (oxyhydr)oxides results in the formation of Fe(II) which can induce the transformation of these iron
Martin Saurer et al.
Science (New York, N.Y.), 365(6458), 1144-1149 (2019-09-14)
Mitochondrial ribosomes (mitoribosomes) are large ribonucleoprotein complexes that synthesize proteins encoded by the mitochondrial genome. An extensive cellular machinery responsible for ribosome assembly has been described only for eukaryotic cytosolic ribosomes. Here we report that the assembly of the small
Veronika Paková et al.
Environmental toxicology and chemistry, 25(12), 3238-3245 (2007-01-16)
N-heterocyclic derivatives of polycyclic aromatic hydrocarbons (NPAHs) are widespread concomitantly with their parent analogues and have been detected in air, water, sediments, and soil. Although they were shown to be highly toxic to some organisms, our understanding of their occurrence
Katsuya Yamada et al.
Mutation research, 559(1-2), 83-95 (2004-04-07)
Phenanthrene, a simplest angular polycyclic aromatic hydrocarbon with a bay-region in its molecule, is reported to be non-mutagenic, although most angular (non-linear) polycyclic aromatic hydrocarbons, such as benzo[a]pyrene and chrysene, are known to show genotoxicity after metabolic transformation into a
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![Benzo[h]quinoline 97%](/deepweb/assets/sigmaaldrich/product/structures/344/715/928932d2-4ca4-4402-b56c-85a80100ce17/640/928932d2-4ca4-4402-b56c-85a80100ce17.png)