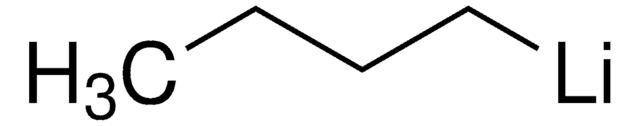
377996
Titanium(IV) isopropoxide
99.999% trace metals basis
Synonym(s):
TTIP, Tetraisopropyl orthotitanate
About This Item
Recommended Products
Quality Level
assay
99.999% trace metals basis
form
liquid
reaction suitability
core: titanium
reagent type: catalyst
refractive index
n20/D 1.464 (lit.)
bp
232 °C (lit.)
mp
14-17 °C (lit.)
density
0.96 g/mL at 20 °C (lit.)
SMILES string
CC(C)O[Ti](OC(C)C)(OC(C)C)OC(C)C
InChI
1S/4C3H7O.Ti/c4*1-3(2)4;/h4*3H,1-2H3;/q4*-1;+4
Looking for similar products? Visit Product Comparison Guide
General description
Application
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Flam. Liq. 3 - STOT SE 3
target_organs
Central nervous system
Storage Class
3 - Flammable liquids
wgk_germany
WGK 1
flash_point_f
105.8 °F - Pensky-Martens closed cup
flash_point_c
41 °C - Pensky-Martens closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Atomic layer deposition (ALD) showcases innovation in novel structure synthesis, area-selective deposition, low-temperature deposition, and more.
From Form to Function: Molding Porous Materials in Three Dimensions by Colloidal Crystal Templating
Reactive silicone chemistry: Focus on pure silicon production, polymerizations, and controlled stereochemistry reactions.
Mesoporous materials, such as aerogels, offer advantages for practical hydrogen storage. They have large surface areas, open porosity, small pore sizes, and the ability to coat the surface with one or more compounds.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service