471933
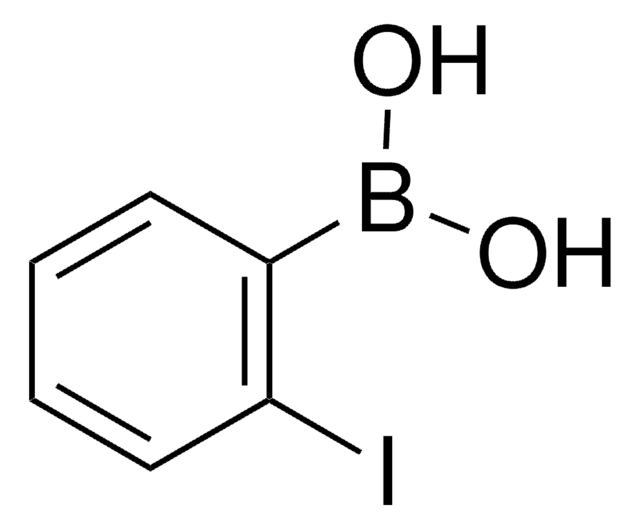
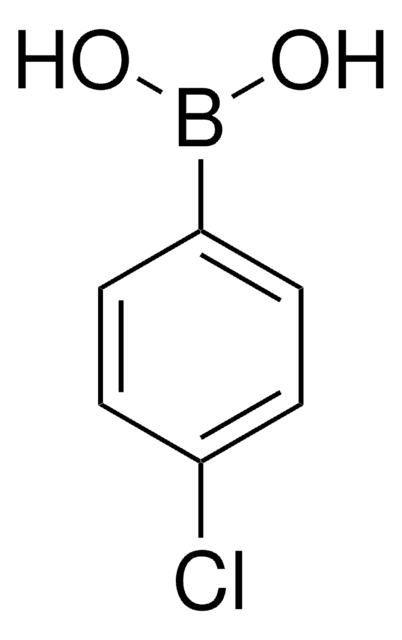
4-Iodophenylboronic acid
≥95.0%
Synonym(s):
B-(4-iodophenyl)-boronic acid, p-Iodophenylboronic acid, p-iodo-benzeneboronic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
IC6H4B(OH)2
CAS Number:
Molecular Weight:
247.83
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
≥95.0%
mp
326-330 °C (lit.)
functional group
iodo
SMILES string
OB(O)c1ccc(I)cc1
InChI
1S/C6H6BIO2/c8-6-3-1-5(2-4-6)7(9)10/h1-4,9-10H
InChI key
PELJYVULHLKXFF-UHFFFAOYSA-N
Related Categories
Application
Reagent used for
Reagent used in Preparation of
- Copper-mediated ligandless aerobic fluoroalkylation
- Palladium-catalyzed aerobic oxidative cross-coupling reactions
- Recyclable magnetic-nanoparticle-supported palladium catalyst for the Suzuki coupling reactions
- Oxidative hydroxylation using a copper (Cu) catalyst
- Ligand-free palladium-catalyzed Suzuki-Miyaura cross-coupling
- Homocoupling using gold salts as a catalyst
- Ruthenium (Ru)-catalyzed cross-coupling
- CuI-catalyzed Suzuki coupling reactions
- Palladium-catalyzed domino Heck-Mizoroki/Suzuki-Miyaura reactions
- Manganese triacetate-mediated radical additions of arylboronic acids to alkenes
Reagent used in Preparation of
- Pleuromutilin derivatives for ribosomal binding and antibacterial activity via "Click Chemistry"
- Liquid crystalline polyacetylene derivatives
Other Notes
Contains varying amounts of anhydride
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
An efficient and recyclable magnetic-nanoparticle-supported palladium catalyst for the Suzuki coupling reactions of organoboronic acids with alkynyl bromides
X. Zhang, et al.,
Synthesis, 18, 2975-2983 (2011)
Arne Dickschat et al.
Organic letters, 12(18), 3972-3974 (2010-08-20)
Arylboronic acids are shown to be valuable precursors for aryl radicals upon treatment with manganese triacetate. Under these oxidative conditions the intermediately generated aryl radicals undergo addition to olefins to give the arylhydroxylation products A in the presence of dioxygen.
Homocoupling of arylboronic acids catalyzed by simple gold salts
Matsuda, T.; et al.
Tetrahedron, 52, 4779-4781 (2011)
Samir Yahiaoui et al.
The Journal of organic chemistry, 76(8), 2433-2438 (2011-03-23)
A palladium(II)-catalyzed Heck-Mizoroki/Suzuki-Miyaura domino reaction involving metal coordinating dimethylaminoethyl vinyl ethers and a number of electron-rich and electron-deficient arylboronic acids has been developed. Through variation of the temperature and the concentration of the p-benzoquinone (p-Bq) ligand/reoxidant, conditions for the robust
Hong Li et al.
Chemical communications (Cambridge, England), 47(27), 7880-7882 (2011-06-11)
The first ruthenium-catalyzed cross-coupling of aldehydes with arylboronic acids is reported. Various aliphatic and aromatic aldehydes are transformed to the corresponding arylketones. A total of 31 examples with moderate to excellent yields are presented, together with the results of an
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service