D194255
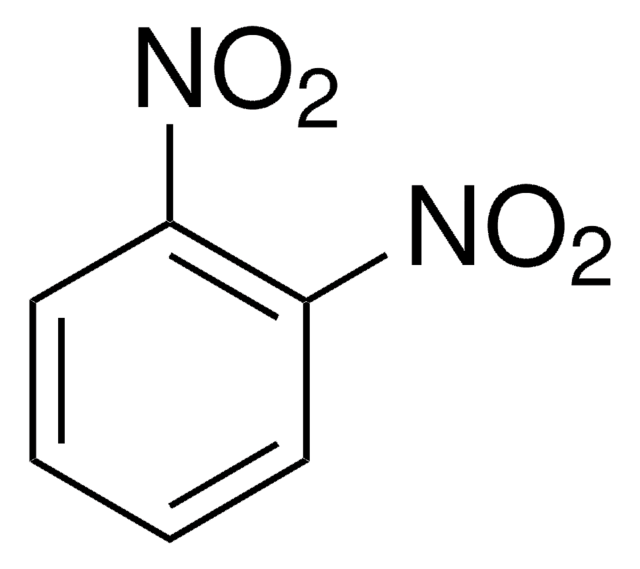
1,3-Dinitrobenzene
97% anhydrous basis
Synonym(s):
1,3-Dinitrobenzene, DNB, Meta-dinitrobenzene, m-Dinitrobenzene
About This Item
Recommended Products
Quality Level
assay
97% anhydrous basis
form
solid
impurities
≤10.0% H2O
bp
297 °C (lit.)
mp
84-86 °C (lit.)
density
1.368 g/mL at 25 °C (lit.)
SMILES string
[O-][N+](=O)c1cccc(c1)[N+]([O-])=O
InChI
1S/C6H4N2O4/c9-7(10)5-2-1-3-6(4-5)8(11)12/h1-4H
InChI key
WDCYWAQPCXBPJA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- 2′,6′- dinitrobiphenyl-4-ol and 1-nitrodibenzofuran via copper-catalyzed regioselective cross-coupling reaction with 4-iodophenol and 2-iodophenol respectively.
- 1H-indazole derivatives by reacting with various N-tosylhydrazones in the presence of a base catalyst.
- 3-(1H-Tetrazol-1-yl)benzenamine by reacting with triethyl orthoformate and NaN3 in the presence of a three-functional redox catalytic system.
signalword
Danger
Hazard Classifications
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - STOT RE 2
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk_germany
WGK 3
flash_point_f
302.0 °F - closed cup
flash_point_c
150 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service