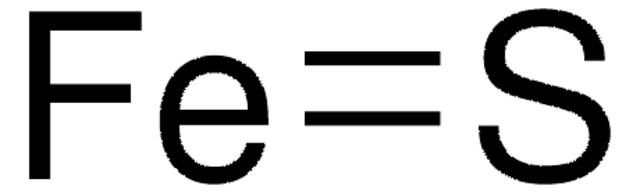15-1520
Iron(II) sulfide
SAJ first grade, ≥50.0%
Synonym(s):
Ferrous sulfide
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
FeS
CAS Number:
Molecular Weight:
87.91
EC Number:
MDL number:
UNSPSC Code:
12352302
PubChem Substance ID:
grade:
SAJ first grade
form:
solid
Recommended Products
grade
SAJ first grade
form
solid
reaction suitability
reagent type: catalyst
core: iron
availability
available only in Japan
concentration
≥50.0%
density
4.84 g/mL at 25 °C (lit.)
SMILES string
S=[Fe]
InChI
1S/Fe.S
InChI key
MBMLMWLHJBBADN-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Elvira Bura-Nakić et al.
Environmental science & technology, 47(2), 741-749 (2012-12-18)
Recent publications have shown that the anodic reaction between FeS and Hg can be used for electrochemical detection of colloidal and particulate FeS in natural waters. Anodic waves that were recorded around -0.45 V (vs Ag/AgCl) in model solutions correspond
Bo Zhang et al.
Proceedings of the National Academy of Sciences of the United States of America, 109(39), 15734-15739 (2012-09-29)
Fumarate and nitrate reduction (FNR) regulatory proteins are O(2)-sensing bacterial transcription factors that control the switch between aerobic and anaerobic metabolism. Under anaerobic conditions [4Fe-4S](2+)-FNR exists as a DNA-binding homodimer. In response to elevated oxygen levels, the [4Fe-4S](2+) cluster undergoes
Chen Xu et al.
ACS nano, 6(6), 4713-4721 (2012-05-10)
We report a facile approach to prepare carbon-coated troilite FeS (C@FeS) nanosheets via surfactant-assisted solution-based synthesis. 1-Dodecanethiol is used as both the sulfur source and the surfactant, which may form different-shaped micelles to direct the growth of nanostructures. Under appropriate
Bin Huang et al.
Bioresource technology, 102(21), 10154-10157 (2011-09-07)
The performance of acetic acid-supported pH-heterogenized heterotrophic denitrification (HD) facilitated with ferrous sulfide-based autotrophic denitrification (AD) was investigated in upflow activated carbon-packed column reactors for reliable removal of highly elevated nitrate (42 mg NO(3)-Nl(-1)) in drinking water. The use of
Dong Suk Han et al.
Journal of hazardous materials, 186(1), 451-457 (2010-11-30)
Higher concentrations (127, 253 μM) of Se(IV) at pH 8 were completely removed by 0.5 g/L FeS within 120 min. Removal of Se(VI) by FeS at pH 8 was less extensive than removal of Se(IV). Only 10% of the Se(VI)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service