86477
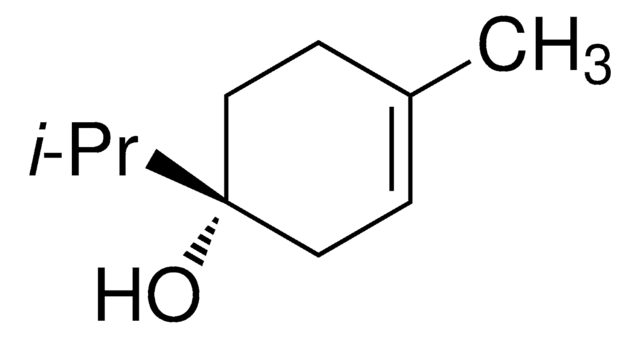
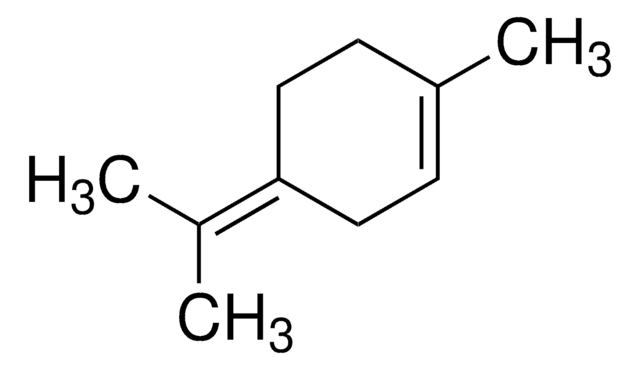
(+)-Terpinen-4-ol
analytical standard
Synonym(s):
(S)-1-Isopropyl-4-methyl-3-cyclohexen-1-ol, (S)-p-Menth-1-en-4-ol
About This Item
Recommended Products
grade
analytical standard
Quality Level
assay
≥98.0% (sum of enantiomers, GC)
form
(Liquid or Viscous Liquid)
optical purity
enantiomeric ratio: ~2:1
shelf life
limited shelf life, expiry date on the label
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
refractive index
n20/D 1.479
bp
211-213 °C (lit.)
density
0.933 g/mL at 20 °C (lit.)
application(s)
food and beverages
format
neat
storage temp.
2-8°C
SMILES string
CC(C)[C@]1(O)CCC(C)=CC1
InChI
1S/C10H18O/c1-8(2)10(11)6-4-9(3)5-7-10/h4,8,11H,5-7H2,1-3H3/t10-/m1/s1
InChI key
WRYLYDPHFGVWKC-SNVBAGLBSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Other Notes
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
174.2 °F - closed cup
flash_point_c
79 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
-β-Farnesene; α-Huµlene; Germacrene D; (+)-Valencene; Bicyclogermacrene; (+)-δ-Cadinene
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service