G4541
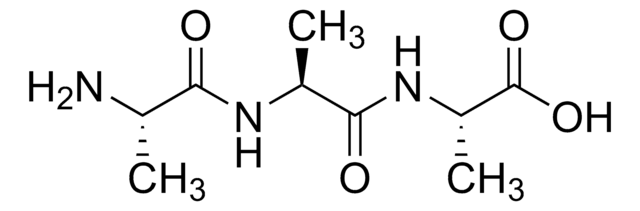
Gly-Gly-His
≥98% (TLC)
Synonym(s):
Copper-binding peptide
About This Item
Recommended Products
Product Name
Gly-Gly-His,
assay
≥98% (TLC)
Quality Level
form
solid
color
white
storage temp.
−20°C
InChI
1S/C10H15N5O4/c11-2-8(16)13-4-9(17)15-7(10(18)19)1-6-3-12-5-14-6/h3,5,7H,1-2,4,11H2,(H,12,14)(H,13,16)(H,15,17)(H,18,19)
InChI key
PDAWDNVHMUKWJR-UHFFFAOYSA-N
Related Categories
Amino Acid Sequence
General description
Application
- as a peptide test probe in the hydrophilic interaction liquid chromatography (HILIC) studies[2]
- in the electrochemical modification of back-side contact transducers (BSC) electrodes in copper (Cu2+) sensing studies[1]
- for the fabrication carbon nanotube electrode for detection of Cu2+ ions[3]
Biochem/physiol Actions
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service