All Photos(1)
About This Item
Empirical Formula (Hill Notation):
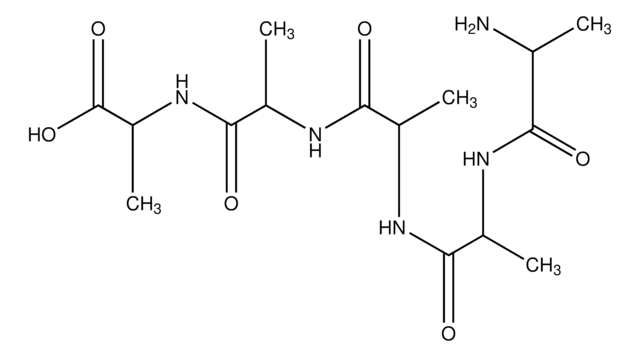
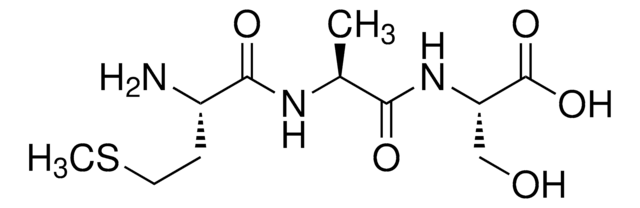
C9H17N3O4
CAS Number:
Molecular Weight:
231.25
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.26
Recommended Products
Product Name
Ala-Ala-Ala,
assay
≥98% (TLC)
Quality Level
form
powder
color
white
storage temp.
−20°C
SMILES string
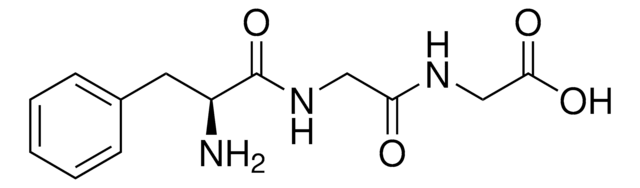
C[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](C)C(O)=O
InChI
1S/C9H17N3O4/c1-4(10)7(13)11-5(2)8(14)12-6(3)9(15)16/h4-6H,10H2,1-3H3,(H,11,13)(H,12,14)(H,15,16)/t4-,5-,6-/m0/s1
InChI key
BYXHQQCXAJARLQ-ZLUOBGJFSA-N
Looking for similar products? Visit Product Comparison Guide
Amino Acid Sequence
Ala-Ala-Ala
Application
Trialanine (Ala-Ala-Ala) may be used along with other short chain alanines, tetra- and penta-alanine, as model compounds to study physicochemical parameters of small peptides.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Fatma Eker et al.
Journal of the American Chemical Society, 125(27), 8178-8185 (2003-07-03)
Determination of the precise solution structure of peptides is of utmost importance to the understanding of protein folding and peptide drugs. Herein, we have measured the UV circular dichroism (UVCD) spectra of tri-alanine dissolved in D(2)O, H(2)O, and glycerol. The
Kaicong Cai et al.
Physical chemistry chemical physics : PCCP, 11(40), 9149-9159 (2009-10-09)
A molecular mechanics (MM) force field-based empirical electrostatic potential map (MM map) for amide-I vibrations is developed with the aim of seeking a quick and reasonable approach to computing local mode parameters and their distributions in solution phase. Using N-methylacetamide
Róisín Moriarty et al.
Thrombosis and haemostasis, 113(2), 290-304 (2014-11-22)
The integrin αIIbβ3 on resting platelets can bind to immobilised fibrinogen resulting in platelet spreading and activation but requires activation to bind to soluble fibrinogen. αIIbβ3 is known to interact with the general integrin-recognition motif RGD (arginine-glycine-aspartate) as well as
Kazuo Yamauchi et al.
Journal of magnetic resonance (San Diego, Calif. : 1997), 190(2), 327-332 (2007-12-07)
(17)O chemical shifts of Ala-Ala-Ala, with parallel and anti-parallel beta-sheet structures, are observed using a 930-MHz high-resolution solid-state NMR spectrometer. Ala-Ala-Ala serves as a model sheet-forming peptide because it can be easily prepared as either a parallel or an anti-parallel
Reinhard Schweitzer-Stenner
The journal of physical chemistry. B, 113(9), 2922-2932 (2009-02-27)
The conformational preference of individual amino acid residues in the unfolded state of peptides and proteins is the subject of a continuous debate. Research has mostly been focused on alanine, owing to its abundance in proteins and its relevance for
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service