D1980000
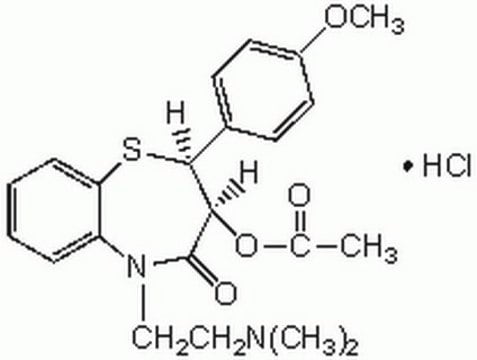
(+)-cis-ジルチアゼム 塩酸塩
European Pharmacopoeia (EP) Reference Standard
別名:
(2S,3S)-(+)-cis-3-アセトキシ-5-(2-ジメチルアミノエチル)-2,3-ジヒドロ-2-(4-メトキシフェニル)-1,5-ベンゾチアゼピン-4(5H)-オン 塩酸塩, CRD-401
ログイン組織・契約価格を表示する
すべての画像(1)
About This Item
実験式(ヒル表記法):
C22H26N2O4S · HCl
CAS番号:
分子量:
450.98
Beilstein:
4228706
MDL番号:
UNSPSCコード:
41116107
PubChem Substance ID:
NACRES:
NA.24
おすすめの製品
グレード
pharmaceutical primary standard
APIファミリー
diltiazem
メーカー/製品名
EDQM
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
SMILES記法
Cl.COc1ccc(cc1)[C@@H]2Sc3ccccc3N(CCN(C)C)C(=O)[C@@H]2OC(C)=O
InChI
1S/C22H26N2O4S.ClH/c1-15(25)28-20-21(16-9-11-17(27-4)12-10-16)29-19-8-6-5-7-18(19)24(22(20)26)14-13-23(2)3;/h5-12,20-21H,13-14H2,1-4H3;1H/t20-,21+;/m1./s1
InChI Key
HDRXZJPWHTXQRI-BHDTVMLSSA-N
遺伝子情報
human ... CACNA1C(775) , CACNA1D(776) , CACNA1F(778) , CACNA1S(779)
類似した製品をお探しですか? 訪問 製品比較ガイド
関連するカテゴリー
詳細
この製品は薬局方標準品です。発行元の薬局方により製造・供給されています。MSDSを含む製品情報などの詳しい情報は、発行元の薬局方のウェブサイトよりご確認ください。
アプリケーション
Diltiazem hydrochloride EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
包装
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
その他情報
Sales restrictions may apply.
関連製品
製品番号
詳細
価格
シグナルワード
Warning
危険有害性情報
危険有害性の分類
Acute Tox. 4 Oral
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
最新バージョンのいずれかを選択してください:
この製品を見ている人はこちらもチェック
Dong Huang et al.
American heart journal, 164(3), 394-401 (2012-09-18)
No randomized trial has been conducted to compare different vasodilators for treating no-reflow during primary percutaneous coronary intervention (PCI) for ST-segment elevation acute myocardial infarction. The prospective, randomized, 2-center trial was designed to compare the effect of 3 different vasodilators
Vida Garaliene et al.
Pharmacological reports : PR, 64(3), 733-738 (2012-07-21)
The goal of this study was to investigate the modulation of the contraction-relaxation effects in isolated human thoracic artery samples of three calcium-channel antagonists, amlodipine (CAS [88150-42-9]), cerebrocrast (CAS [118790-71-9]) and diltiazem (CAS [42399-41-7]), and two calcium-channel agonists, CGP 28392
Andrew P Ambrosy et al.
The American journal of cardiology, 110(11), 1623-1626 (2012-09-04)
Five patients with primary colorectal adenocarcinoma or anal squamous cell carcinoma were started on a 2-weeks-on, 1-week-off capecitabine dosing regimen in addition to other chemotherapeutic agents and/or radiation. Within the first few doses, patients experienced chest pain and/or dyspnea at
Lun Li et al.
PloS one, 7(6), e38851-e38851 (2012-07-05)
The aim of this study is to observe the chronic effects of diltiazem release capsules on patients with coronary slow flow (CSF) phenomenon. From 2004 to 2009, 80 consecutive patients with chest pain and normal coronary arteries evidenced by coronary
Mustafa Kayan et al.
The Journal of membrane biology, 245(12), 833-840 (2012-08-21)
Non-ionic contrast media (CM) can induce tissue kidney injury via activation of phagocytosis and oxidative stress, although the mechanisms of injury via neutrophils are not clear. We investigated the effects of CM on oxidative stress and Ca²⁺ concentrations in serum
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)