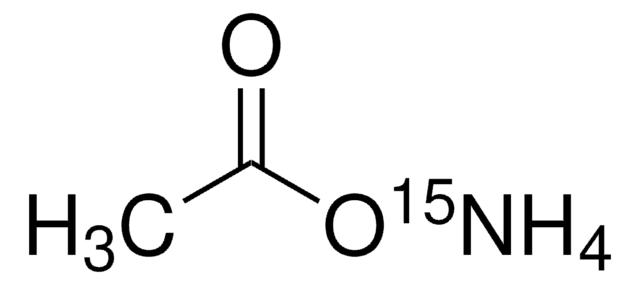
おすすめの製品
形状
liquid
包装
pkg of 10 μg (3 vials)
メーカー/製品名
NIST®
アプリケーション
genomic analysis
保管温度
−20°C
詳細
Human DNA for Whole-Genome Variant Assessment Reference Material (RM) is intended for validation, optimization, and process evaluation purposes. It consists of three human whole genome samples from a son-father-mother family trio of Eastern European Ashkenazi Jewish ancestry from the Personal Genome Project (IDs huAA53E0, hu6E4515, and hu8E87A9). A unit of RM 8392 consists of three vials containing human genomic DNA from a specific family member; extracted from three large growths of human lymphoblastoid cell lines from the Coriell Institute for Medical Research (Camden, NJ): GM24385 (son) labeled as HG-002, GM24149 (father) labeled as HG-003, and GM24143 (mother) labeled as HG-004. Each vial contains approximately 10 µg of genomic DNA, and the DNA is in TE buffer (10 mM TRIS, 1 mM EDTA, pH 8.0)
SRM 8392_cert
SRM 8392_SDS
SRM 8392_cert
SRM 8392_SDS
アプリケーション
Human DNA for Whole-Genome Variant Assessment Reference Material is intended for assessing the performance of human genome sequencing variant calling by obtaining estimates of true positives, false positives, and false negatives. Sequencing applications could include:
- whole genome sequencing
- whole exome sequencing
- targeted sequencing such as gene panels
特徴および利点
- This reference material contains isolated DNA rather than live cells and is intended for research use.
- An extensive report of the investigation is available through NIST
- Information values are provided for single nucleotide variations (SNVs), small insertions and deletions (indels), and homozygous reference genotypes.
その他情報
- RM 8392 is stored at –20 °C at NIST but will be shipped in freezer packs and may not arrive frozen.
- Details on expiration, storage, safety, usage, and source are provided in the NIST certificate.
- Information on biomaterials, disposal, and transport is available in the SDS.
- he size distributions are measured using PFGE, and biases of this method were not characterized.
法的情報
NIST is a registered trademark of National Institute of Standards and Technology
SRM is a registered trademark of National Institute of Standards and Technology
保管分類コード
12 - Non Combustible Liquids
WGK
WGK 1
引火点(°F)
Not applicable
引火点(℃)
Not applicable
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
NIST8392:
NIST8393-10EA:
最新バージョンのいずれかを選択してください:
Determining Performance Metrics for Targeted Next-Generation Sequencing Panels Using Reference Materials
Cleveland MH, et al.
The Journal of Molecular Diagnostics : JMD, 20, 583-590 (2018)
Bennett O V Shum et al.
The Journal of molecular diagnostics : JMD, 19(4), 602-612 (2017-05-16)
The sensitivity and specificity of next-generation sequencing laboratory developed tests (LDTs) are typically determined by an analyte-specific approach. Analyte-specific validations use disease-specific controls to assess an LDT's ability to detect known pathogenic variants. Alternatively, a methods-based approach can be used
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)