PHR1308
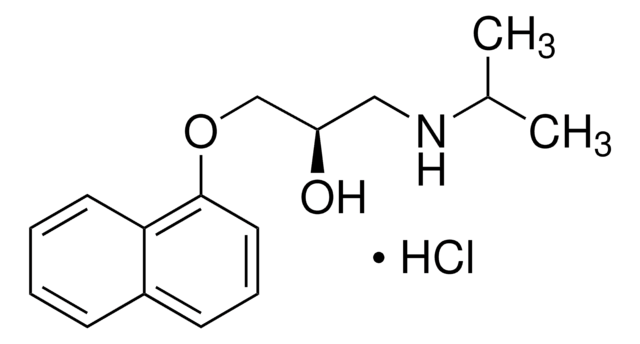
(±)-プロプラノロール 塩酸塩
Pharmaceutical Secondary Standard; Certified Reference Material
別名:
(±)-1-イソプロピルアミノ-3-(1-ナフチロキシ)-2-プロパノール 塩酸塩, DL-プロプラノロール 塩酸塩
About This Item
おすすめの製品
グレード
certified reference material
pharmaceutical secondary standard
品質水準
認証
traceable to Ph. Eur. P3500000
traceable to USP 1576005
APIファミリー
propranolol
CofA
current certificate can be downloaded
テクニック
HPLC: suitable
gas chromatography (GC): suitable
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
保管温度
2-30°C
SMILES記法
Cl.CC(C)NCC(O)COc1cccc2ccccc12
InChI
1S/C16H21NO2.ClH/c1-12(2)17-10-14(18)11-19-16-9-5-7-13-6-3-4-8-15(13)16;/h3-9,12,14,17-18H,10-11H2,1-2H3;1H
InChI Key
ZMRUPTIKESYGQW-UHFFFAOYSA-N
遺伝子情報
human ... ADRB1(153) , ADRB2(154)
類似した製品をお探しですか? 訪問 製品比較ガイド
詳細
Propranolol is a β-adrenergic blocking compound, widely used for the management of cardiac arrhythmia, sinus tachycardia, angina pectoris and hypertension. It is also prescribed for the treatment of dysfunctional labor, migraine, as well as anxiety.
アプリケーション
アナリシスノート
その他情報
脚注
おすすめ製品
関連製品
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
PHR1308-1G:
PHR1308-1G-PW:
この製品を見ている人はこちらもチェック
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)