223689
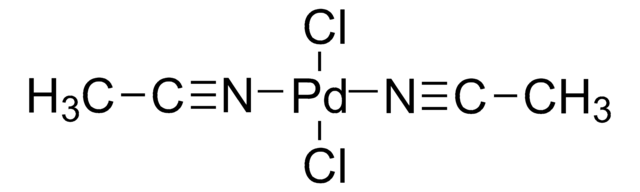
Bis(benzonitrile)palladium(II) chloride
95%
동의어(들):
Benzonitrile, palladium complex, Bis(benzonitrile)dichloropalladium(II), Bis(benzonitrile)palladium(II) chloride, Bis(phenylnitrile)dichloropalladium, Dibenzonitrilepalladium dichloride, Dichlorobis(benzonitrile)palladium, Dichlorobis(benzonitrile)palladium(II), Dichlorobis(phenyl cyanide)palladium, Palladium dichloride bis(benzonitrile), Palladium(II) chloride bis(benzonitrile) complex
About This Item
추천 제품
Quality Level
분석
95%
형태
powder
반응 적합성
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
환경친화적 대안 제품 특성
Catalysis
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
mp
131 °C (lit.)
환경친화적 대안 카테고리
SMILES string
Cl[Pd]Cl.N#Cc1ccccc1.N#Cc2ccccc2
InChI
1S/2C7H5N.2ClH.Pd/c2*8-6-7-4-2-1-3-5-7;;;/h2*1-5H;2*1H;/q;;;;+2/p-2
InChI key
WXNOJTUTEXAZLD-UHFFFAOYSA-L
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
We are committed to bringing you Greener Alternative Products, which adhere to one or more of The 12 Principles of Greener Chemistry. This product has been enhanced for catalytic efficiency. Find details here.
애플리케이션
- For greener amine synthesis from terminal olefins by Wacker oxidation, followed by transfer hydrogenation of the resultant imine.
- In cross-coupling reactions and α-O-glycosidation.
Formal anti-Markovnikov hydroamination of terminal olefins
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.


![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)