추천 제품
Quality Level
분석
98%
mp
100-103 °C (lit.)
작용기
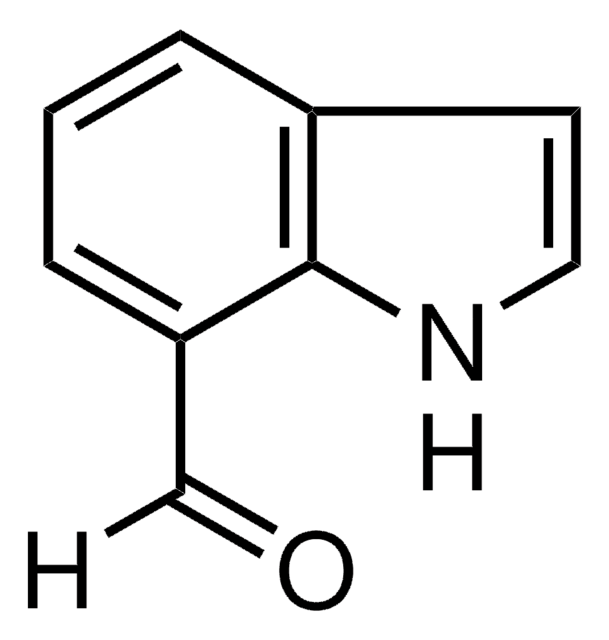
aldehyde
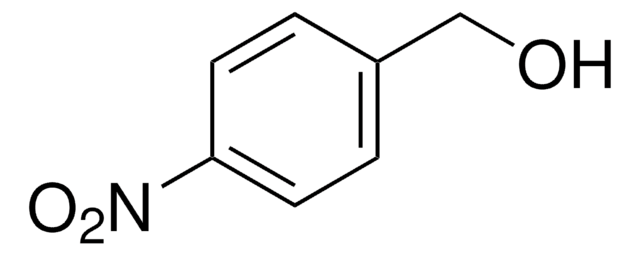
SMILES string
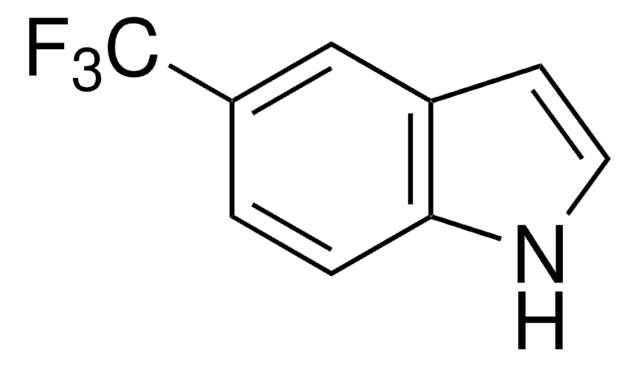
O=Cc1ccc2[nH]ccc2c1
InChI
1S/C9H7NO/c11-6-7-1-2-9-8(5-7)3-4-10-9/h1-6,10H
InChI key
ADZUEEUKBYCSEY-UHFFFAOYSA-N
애플리케이션
Indole-5-carboxaldehyde can be used as a reactant in the:
- Preparation of curcumin derivatives as anti-proliferative & anti-inflammatory agents
- Preparation of analogs of botulinum neurotoxin serotype A protease inhibitors
- Stereoselective synthesis of dibenzylideneacetone derivatives as β-amyloid imaging probes
- Synthesis of para-para stilbenophanes by McMurry coupling
- Stereoselective synthesis of heteroaromatic (E)-α,β-unsaturated ketones from aldehydes
- Structure-based drug design of aurora kinase A inhibitors
- Preparation of 5-indolyl linked 15- and 18-membered azacrown ethers to study their cation-π interactions.
- Preparation of bibenzimidazole derivatives substituted 5-indolyl moiety in the study of inhibition of topoisomerase I activity.
- Synthesis of (5-(4-(3,4,5-trimethoxybenzoyl)-1H-imidazol-2-yl)-1H-indol-2-yl)(3,4,5-trimethoxyphenyl)methanone and radioiodinated indolochalcone.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Todd J Eckroat et al.
Beilstein journal of organic chemistry, 9, 1012-1044 (2013-06-15)
The number of people suffering from Alzheimer's disease (AD) is expected to increase dramatically in the coming years, placing a huge burden on society. Current treatments for AD leave much to be desired, and numerous research efforts around the globe
S Jin et al.
Bioorganic & medicinal chemistry letters, 10(8), 719-723 (2000-04-27)
A series of 2'-heterocyclic derivatives of 5-phenyl-2,5'-1H-bibenzimidazoles were evaluated for topoisomerase I poisoning activity and cytotoxicity. Topo I poisoning activity was associated with 2'-derivatives that possessed a hydrogen atom capable of hydrogen bond formation, suggesting that the interatomic distances between
Synthesis, QSAR and hypoglycemic activity of substituted 2,4-thiazolidinedione derivatives
Bahara, R. S.; Kulkarni, V. M.
Der Pharma Chemica, 3, 164-164 (2011)
Raquel Álvarez et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 17(12), 3406-3419 (2011-02-24)
The synthesis of a new family of methoxy-substituted [2.7]- and [2.8]paracyclophanes linked by 3-oxapentamethylene-1,5-dioxy and hexamethylene-1,6-dioxy bridges has been carried out by using the McMurry methodology. Related indole compounds were also synthesised. Olefin-to-diol ratios depended on the bridge length, the
Houlihan WJ.
The Chemistry of Heterocyclic Compounds, 367-367 (2009)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.