추천 제품
vapor density
1.93 (vs air)
Quality Level
vapor pressure
11.6 mmHg ( 20 °C)
분석
99%
refractive index
n20/D 1.432 (lit.)
bp
114-115 °C (lit.)
mp
−53 °C (lit.)
density
0.963 g/mL at 25 °C (lit.)
저장 온도
2-8°C
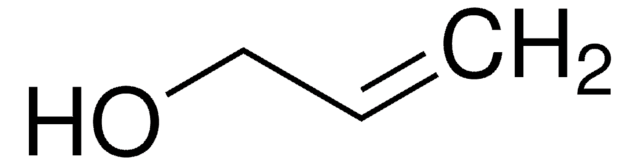
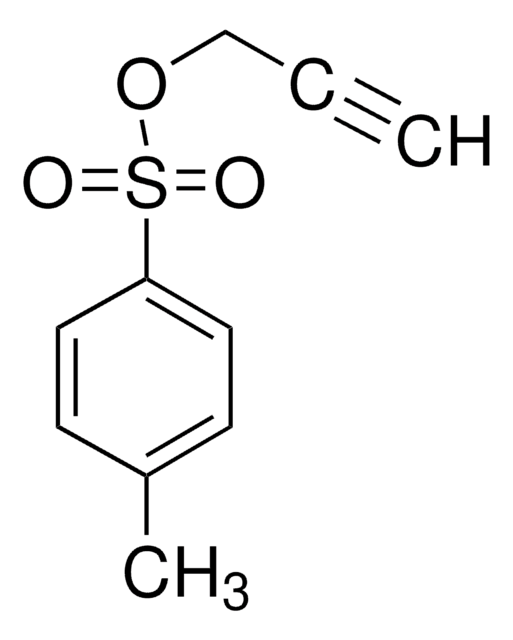
SMILES string
OCC#C
InChI
1S/C3H4O/c1-2-3-4/h1,4H,3H2
InChI key
TVDSBUOJIPERQY-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
Propargyl alcohol has been used as a key starting material in the [4+2] cycloisomerization mediated synthesis of various phthalide derivatives.
It can also be used to synthesize:
It can also be used to synthesize:
- A variety of regioselective furan-3-carboxamides by reacting with 3-oxo amides using Ag2CO3 as a promoter.
- β-oxopropyl esters by reacting with carboxylic acids in the presence of (arene) (phosphine)ruthenium(II) complex as a catalyst.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 2 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Chronic 2 - Eye Dam. 1 - Flam. Liq. 3 - Skin Corr. 1B - STOT RE 2
표적 기관
Liver,Kidney
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point (°F)
91.4 °F - closed cup
Flash Point (°C)
33 °C - closed cup
개인 보호 장비
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
이미 열람한 고객
Guangyan Zhang et al.
Polymers, 11(2) (2019-04-10)
The temperature responsive PEGylated polyaspartamide derivative, denoted as mPEG-PAAHP, was synthesized by the click reaction. FTIR and ¹H NMR were adopted to characterize and confirm the chemical structures of the obtained mPEG-PAAHPs. The temperature responsive behavior investigated by transmittance and
A Stephen K Hashmi et al.
The Journal of organic chemistry, 77(17), 7761-7767 (2012-08-11)
The gold(I)-catalyzed oxidative rearrangement of propargyl alcohols provides an efficient and selective route to 1,3-diketones under mild conditions. Pyridine-N-oxides were used as external oxidants with, different from related substrates, no alkylidenecycloalkanones or oxetan-3-ones formed as side-products.
Ming Chen et al.
Journal of the American Chemical Society, 134(26), 10947-10952 (2012-06-27)
Chiral Brønsted acid catalyzed asymmetric allenylboration reactions are described. Under optimized conditions, anti-homopropargyl alcohols 2 are obtained in high yields with excellent diastereo- and enantioselectivities from stereochemically matched aldehyde allenylboration reactions with (M)-1 catalyzed by the chiral phosphoric acid (S)-4.
Jin Kyoon Park et al.
Organic letters, 14(18), 4790-4793 (2012-09-06)
The catalytic regioselective hydroboration of propargylic alcohols and ethers was investigated using NHC-CuCl. We observe that different NHC-CuCl complexes catalyze hydroborations of propargylic substrates with opposite regioselectivity. A 6-NHC-CuCl complex provides α-selectivity whereas β-selectivity is achieved using a 5-NHC-CuCl complex.
Hua-Dong Xu et al.
Organic letters, 15(4), 840-843 (2013-01-30)
Cross-dimerization of terminal arylacetylenes and terminal propargylic alcohols/amides has been achieved in the presence of a rhodium catalyst. This method features high chemo- and regioselectivities rendering convenient and atom economical access to functionalized enynes.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.