추천 제품
Grade
pharmaceutical primary standard
API family
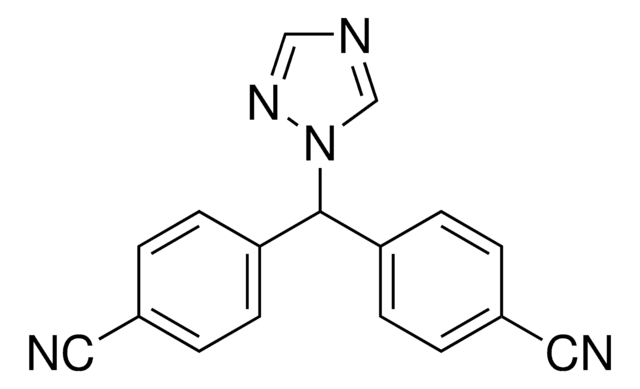
letrozole
제조업체/상표
EDQM
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-8°C
SMILES string
N#CC(C=C1)=CC=C1C(N2C=NC=N2)C3=CC=C(C#N)C=C3
InChI
1S/C17H11N5/c18-9-13-1-5-15(6-2-13)17(22-12-20-11-21-22)16-7-3-14(10-19)4-8-16/h1-8,11-12,17H
InChI key
HPJKCIUCZWXJDR-UHFFFAOYSA-N
유전자 정보
human ... CYP19A1(1588)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the Issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
애플리케이션
Letrozole EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
생화학적/생리학적 작용
Letrozole is a non-steroidal aromatase inhibitor.
Letrozole is a third generation nonsteroidal aromatase inhibitor. It is a competitive inhibitor of the aromatase enzyme system and thus inhibits the conversion of androgens to estrogens. Letrozole inhibits the aromatase enzyme by competitively binding to the heme of the cytochrome P450 subunit of the enzyme, resulting in a reduction of estrogen biosynthesis in all tissues.
포장
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
기타 정보
Sales restrictions may apply.
관련 제품
제품 번호
설명
가격
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Repr. 2 - STOT RE 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
Antonio C Wolff et al.
Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 31(2), 195-202 (2012-12-13)
Recent data showed improvement in progression-free survival (PFS) when adding everolimus to exemestane in patients with advanced breast cancer experiencing recurrence/progression after nonsteroidal aromatase inhibitor (AI) therapy. Here, we report clinical outcomes of combining the mammalian target of rapamycin (mTOR)
Valentina Guarneri et al.
Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 32(10), 1050-1057 (2014-03-05)
This is a randomized, double-blind, placebo-controlled study aimed to evaluate the clinical and biologic effects of letrozole plus lapatinib or placebo as neoadjuvant therapy in hormone receptor (HR) -positive/human epidermal growth factor receptor 2 (HER2) -negative operable breast cancer. Ninety-two
G Allevi et al.
British journal of cancer, 108(8), 1587-1592 (2013-04-13)
The objective of this study was to determine the optimal scheduling of 2.5 mg daily letrozole in neoadjuvant breast cancer patients to obtain pathological complete response (pathCR) and assess Ki-67 expression as an early predictor of response. This single institution
Mothaffar F Rimawi et al.
Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 31(14), 1726-1731 (2013-04-10)
We previously reported the eradication of human epidermal growth factor receptor 2 (HER2)- amplified human xenografts in mice by inhibition of the HER2 pathway with lapatinib and trastuzumab to block all homo- and heterodimer signaling as well as by blockade
Dennis C Sgroi et al.
Journal of the National Cancer Institute, 105(14), 1036-1042 (2013-07-03)
Biomarkers to optimize extended adjuvant endocrine therapy for women with estrogen receptor (ER)-positive breast cancer are limited. The HOXB13/IL17BR (H/I) biomarker predicts recurrence risk in ER-positive, lymph node-negative breast cancer patients. H/I was evaluated in MA.17 trial for prognostic performance
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.