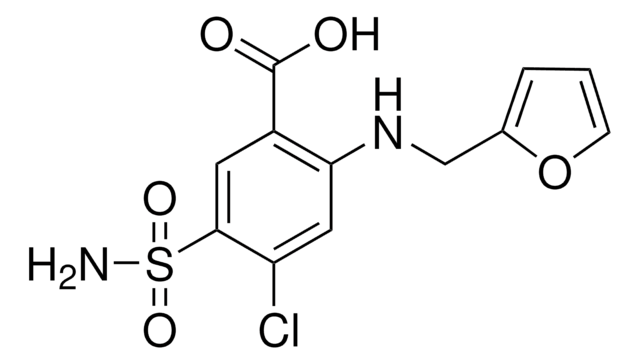
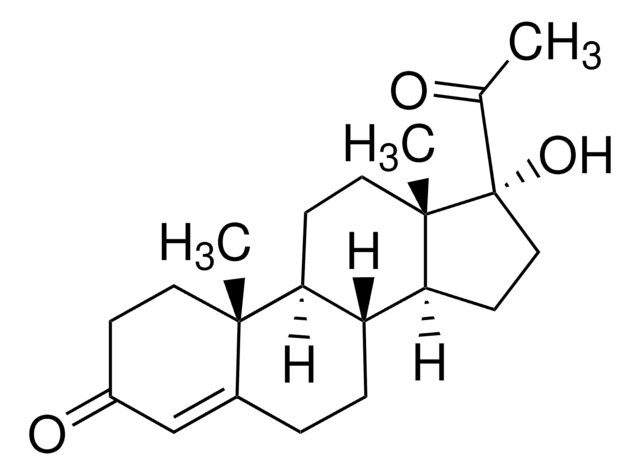
D4875
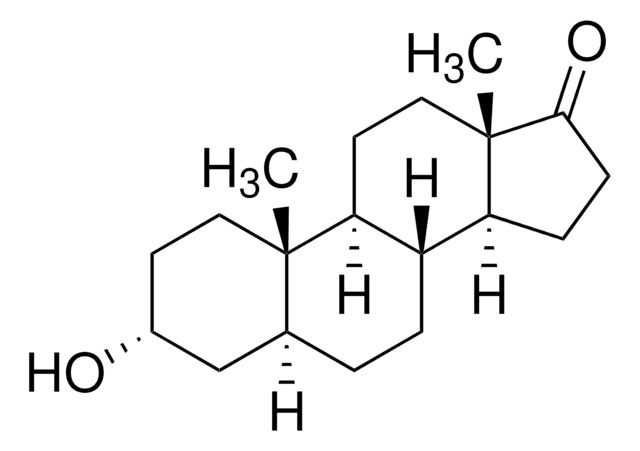
16-Dehydropregnenolone acetate
≥95%
동의어(들):
3β-Acetoxy-5,16-pregnadien-20-one, 3β-Hydroxy-5,16-pregnadien-20-one acetate, 5,16-Pregnadien-3β-ol-20-one acetate
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C23H32O3
CAS Number:
Molecular Weight:
356.50
EC Number:
MDL number:
UNSPSC 코드:
12352202
PubChem Substance ID:
NACRES:
NA.77
추천 제품
분석
≥95%
형태
powder
SMILES string
CC(=O)OC1CCC2(C)C3CCC4(C)C(CC=C4C(C)=O)C3CC=C2C1
InChI
1S/C23H32O3/c1-14(24)19-7-8-20-18-6-5-16-13-17(26-15(2)25)9-11-22(16,3)21(18)10-12-23(19,20)4/h5,7,17-18,20-21H,6,8-13H2,1-4H3
InChI key
MZWRIOUCMXPLKV-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
생화학적/생리학적 작용
16-Dehydropregnenolone acetate (DPA) is synthesized from steroids sapogenin, diosgenin and solasodine. 16-Dehydropregnenolone acetate (DPA) is a crucial intermediate for the synthesis of steroid hormones-based drugs. It is an antagonist for farnesoid X receptor (FXR) and modulates cholesterol metabolism. It is considered as a potential antihyperlipidemic agent. Chemically synthesized steroid derivatives from DPA have cytotoxic features and could serve as potential anticancer agents.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
S B Mahato et al.
The Biochemical journal, 255(3), 769-774 (1988-11-01)
Metabolic processes involving side-chain and ring cleavage of progesterone, 17-hydroxyprogesterone, 11-deoxycortisol and 16-dehydropregnenolone by Arthrobacter simplex were studied. The formation of the metabolites from progesterone indicates a pathway somewhat different from normal in the enzymic reaction sequence, and the 17-hydroxyprogesterone
A one-pot efficient process for 16-dehydropregnenolone acetate
Goswami A, et al.
Organic Process Research & Development, 7(3), 306-308 (2003)
Víctor Pérez-Ornelas et al.
Steroids, 70(3), 217-224 (2005-03-15)
The enzyme 5alpha-reductase is responsible for the conversion of testosterone (T) to its more potent androgen dihydrotestosterone (DHT). This steroid had been implicated in androgen-dependent diseases such as: benign prostatic hyperplasia, prostate cancer, acne and androgenic alopecia. The inhibition of
A V Kamernitzky et al.
Journal of steroid biochemistry, 16(1), 61-67 (1982-01-01)
A new class of modified progesterones with an additional ring in the 16 alpha , 17 alpha-position (pregna-D'-pentaranes) are described. Compounds containing 4- and 6-membered D'-ring (D'4- and D'6-pentaranes) were synthesized by the cycloaddition of acetylene or 1,3-butadiene, respectively, to
16-Dehydropregnenolone lowers serum cholesterol by up-regulation of CYP7A1 in hyperlipidemic male hamsters
Ramakrishna R, et al.
The Journal of Steroid Biochemistry and Molecular Biology, 168(1), 110-117 (2017)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.