241059
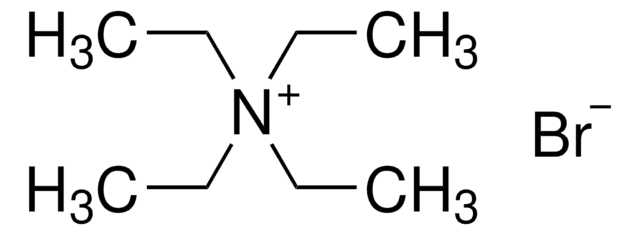
Tetraethylammonium bromide
ReagentPlus®, 99%
Synonym(s):
TEAB, TEA bromide
About This Item
Recommended Products
grade
reagent
product line
ReagentPlus®
assay
99%
form
crystals
dilution
(for general lab use)
impurities
1% triethylamine hydrobromide
pH
6.5 (100 g/L)
mp
285 °C (dec.) (lit.)
SMILES string
[Br-].CC[N+](CC)(CC)CC
InChI
1S/C8H20N.BrH/c1-5-9(6-2,7-3)8-4;/h5-8H2,1-4H3;1H/q+1;/p-1
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- Tetraethylammonium bromide (TEAB) catalyzes the synthesis of thioesters by the oxidative coupling of aldehydes or alcohols with thiols or disulfides.[1]
- It is used as a catalyst, along with o-iodoxybenzoic acid (IBX), in the oxidation of sulfides to sulfoxides[2] and primary carboxamides to one-carbon dehomologated nitriles.[3]
- TEAB can also be used as an organic template to synthesize zeolite beta.[4]
Legal Information
hcodes
pcodes
Hazard Classifications
Aquatic Chronic 3
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
361.4 °F - closed cup
flash_point_c
183 °C - closed cup
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service