441252
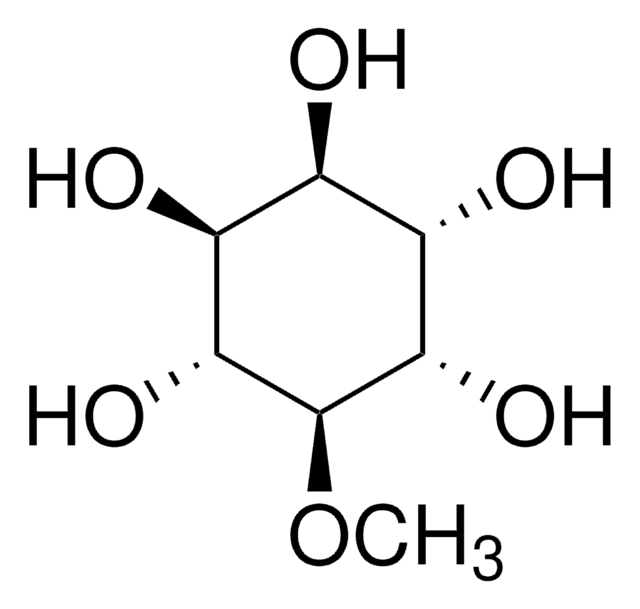
D-Pinitol
95%
Synonym(s):
3-O-Methyl-D-chiro-inositol
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C7H14O6
CAS Number:
Molecular Weight:
194.18
MDL number:
UNSPSC Code:
12352112
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
95%
form
powder
optical activity
[α]20/D 60.0 to 70.0°, c = 1% in H2O
mp
179-185 °C (lit.)
functional group
ether
hydroxyl
SMILES string
CO[C@@H]1[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)[C@H]1O
InChI
1S/C7H14O6/c1-13-7-5(11)3(9)2(8)4(10)6(7)12/h2-12H,1H3/t2-,3-,4-,5-,6+,7+/m0/s1
InChI key
DSCFFEYYQKSRSV-KLJZZCKASA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
D-pinitol, commonly found conifers, is an isomer of L-quebrachitol.
Application
D-pinitol may be used as a starting material to prepare its azole nucleoside analogs. It may also be used in the preparation of 1D-1,5-dideoxy-1,5-difluoro-neo-inositol and 1D-1-deoxy-1-fluoro-myo-inositol.
Precursor to biologically active fluorinated isosteres of inositol, which show cell growth inhibitory properties. Has shown antidiabetic properties in mice. Believed to be a salt stress regulator in a wide range of plants.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
A Llanes et al.
Plant biology (Stuttgart, Germany), 15 Suppl 1, 118-125 (2012-07-04)
The success of Prosopis strombulifera in growing under high NaCl concentrations involves a carefully controlled balance among different processes, including compartmentation of Cl(-) and Na(+) in leaf vacuoles, exclusion of Na(+) in roots, osmotic adjustment and low transpiration. In contrast
Shan-Chi Liu et al.
International immunopharmacology, 12(3), 494-500 (2012-01-25)
Numerous studies have indicated that inflammatory cytokines play a major role in osteoclastogenesis, leading to the bone resorption that is frequently associated with osteoporosis. D-pinitol, a 3-methoxy analogue of D-chiroinositol, was identified as an active principle in soy foods and
Synthesis of azole nucleoside analogues of D-pinitol as potential antitumor agents.
Zhan T and Lou H.
Carbohydrate Research, 342(6), 865-869 (2007)
Nhung Thuy Dang et al.
Bioscience, biotechnology, and biochemistry, 74(5), 1062-1067 (2010-05-13)
Diabetes mellitus is a complex disease that is characterized by the defection of insulin sensitivity in such peripheral tissues as skeletal muscle, adipose tissue and liver. We have previously demonstrated that certain inositol derivatives stimulated glucose uptake accompanied by the
J. C. Thomas et al.
Plant physiology, 103(4), 1299-1304 (1993-12-01)
We selected indicators of four different metabolic processes (Crassulacean acid metabolism [CAM], amino acid and nitrogen mobilization metabolism, osmoprotection, and plant defense mechanisms) to study the relationship between salt-stress-mediated and plant growth regulator (PGR)-induced responses in Mesembryanthemum crystallinum (ice plant).
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service