218783
Caspase-1, Human, Recombinant, E. coli
Caspase-1, Human, Recombinant, is expressed in E. coli and is fused at the N-terminus to a His•Tag sequence.
Synonym(s):
IL-1β Converting Enzyme, ICE
About This Item
Recommended Products
recombinant
expressed in E. coli
Quality Level
assay
≥90% (SDS-PAGE)
form
liquid
specific activity
≥25,000 units/mg protein
≥50,000 units/mL
manufacturer/tradename
Calbiochem®
storage condition
OK to freeze
avoid repeated freeze/thaw cycles
shipped in
dry ice
storage temp.
−70°C
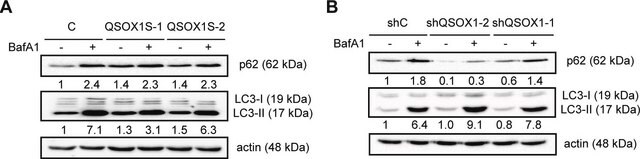
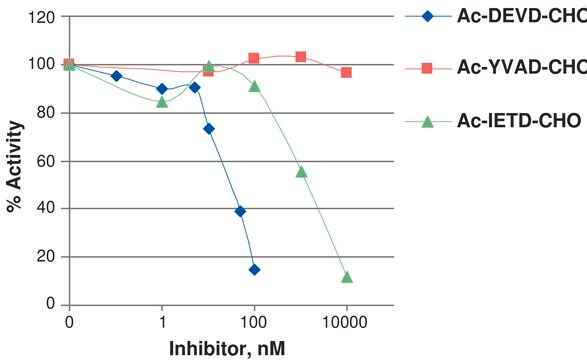
General description
Caspases are enzymes that belong to the cysteine protease family. [1] In mammals, researchers have discovered 13 caspases. [2] They enhance chemical reactions by targeting specific substances, guided by aspartic acid. These caspases can be further classified into two groups: apoptotic caspases (caspase-2, 3, 6, 7, 8, 9, and 10) and inflammatory caspases (caspase-1, 4, 5, 11, and 12). [1] Apart from inflammatory organs such as the spleen, lymph nodes, and thymus, caspase-1 is also expressed in adipose tissue, liver, and intestine. [3] Recombinant, human caspase-1 fused at the N-terminus to a His•Tag sequence and expressed in E. coli. Useful for the study of enzyme regulation, cleavage of target substrates, and inhibitor screening. M.W. 10000 and 20000.
Application
Biochem/physiol Actions
Warning
Unit Definition
Physical form
Reconstitution
Other Notes
Thornberry, N.A., et al. 1992. Nature 356, 768.
Legal Information
Storage Class
10 - Combustible liquids
wgk_germany
WGK 2
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service