C3556
Catalase from human erythrocytes
≥90% (SDS-PAGE), buffered aqueous solution, ≥30,000 units/mg protein
Synonym(s):
H2O2:H2O2 oxidoreductase
About This Item
Recommended Products
General description
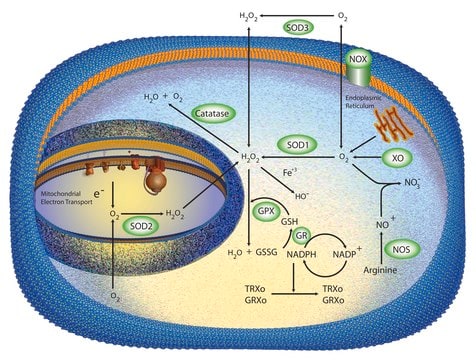
Human catalase is a member of the monofunctional heme-containing catalases. It is an intracellular enzyme located at higher concentrations in the liver, erythrocytes, and kidney. Catalase is a homo-tetrameric protein and comprises amino acid residues, one heme group that is iron III protoporphyrin IX, and a nicotinamide adenine dinucleotide phosphate (NADPH) molecule.[1] It is a ubiquitous[2] enzyme found in most aerobic organisms.[3] The catalase (CAT) gene is located on the human chromosome at 11p13.[4]
Application
- to prevent reoxidation of reduced cytochrome c by H2O2 while measuring the production of superoxide with cytochrome C.[5]
- as a component of the imaging buffer for stochastic optical reconstruction microscopy (STORM) imaging of human skin fibroblasts[6]
- as a component of the gloxy mix for single-molecule imaging[7]
Biochem/physiol Actions
Unit Definition
Physical form
Storage and Stability
persons
Analysis Note
inhibitor
signalword
Danger
hcodes
pcodes
Hazard Classifications
Resp. Sens. 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Cellular oxidative stress is countered by enzymatic scavengers and antioxidant modulators against reactive oxygen species damage.
Protocols
This procedure may be used for all Catalase products.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service