C6854
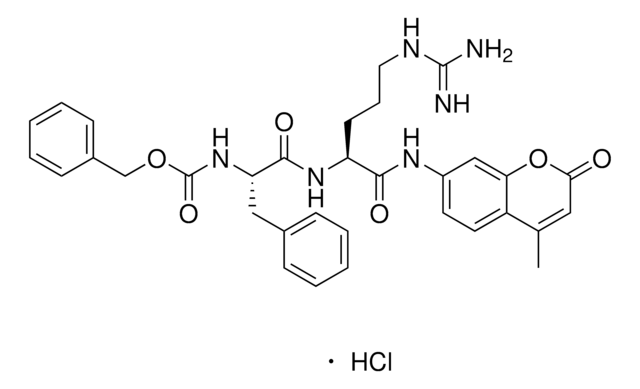
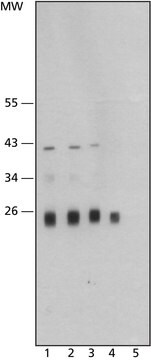
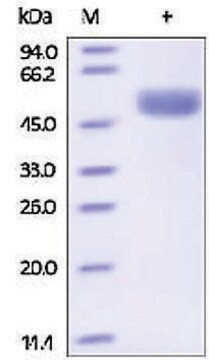
Cathepsin L from human liver
≥0.5 units/mg protein, solution
About This Item
Recommended Products
General description
Application
- to digest mutant transforming growth factor β-induced (TGFBIp) and LC3 (autophagosomes marker) proteins in vitro
- to digest the pH-dependent fibril of a pre-melanosomal protein (Pmel17) repeat domain (RPT) isoform and study the effect of pH on sRPT aggregation kinetics by tryptophan fluorescence
- to analyze the inhibition of CTSL by small molecules or drugs in a cell-free system
Biochem/physiol Actions
Unit Definition
Physical form
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service