おすすめの製品
グレード
certified reference material
pharmaceutical secondary standard
品質水準
認証
traceable to Ph. Eur. B1220000
APIファミリー
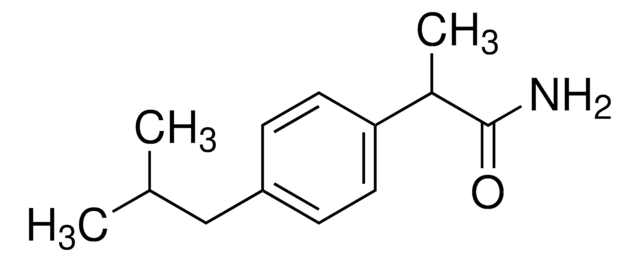
ibuprofen
CofA
current certificate can be downloaded
包装
pkg of 20 mg
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
保管温度
2-8°C
詳細
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
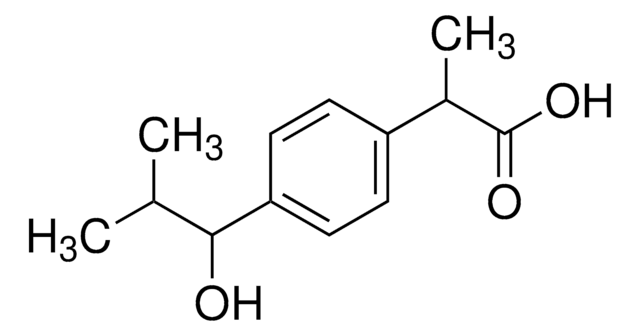
It is an impurity of the nonsteroidal anti-inflammatory drug (NSAID)― ibuprofen, used for the treatment of mild and moderate pain such as rheumatoid arthritis, osteoarthritis, and dysmenorrhea.
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
It is an impurity of the nonsteroidal anti-inflammatory drug (NSAID)― ibuprofen, used for the treatment of mild and moderate pain such as rheumatoid arthritis, osteoarthritis, and dysmenorrhea.
アプリケーション
This pharmaceutical secondary standard can also be used as follows:
- Development of a reverse-phase ultraperformance liquid chromatographic (RP-UPLC) method for the estimation of ibuprofen and diphenhydramine citrate along with their related impurities in their combined dosage form
- Simultaneous determination of ibuprofen and its 17 related impurities by an ICH validated reversed-phase high-performance liquid chromatography (RP-HPLC) method in tablets
アナリシスノート
このような2次標準は、入手可能な場合にはUSP、EP、BPの1次標準にマルチトレーサビリティを提供します。
脚注
To see an example of a Certificate of Analysis for this material enter LRAB8256 in the Documents slot below. This is an example certificate only and may not be the lot that you receive.
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
PHR1934-20MG-PW:
PHR1934-20MG:
この製品を見ている人はこちらもチェック
Development and validation of an HPLC method for simultaneous determination of ibuprofen and 17 related compounds
Han Z, et al.
Chromatographia, 80, 1353-1360 (2017)
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)