おすすめの製品
アッセイ
≥98% (HPLC)
フォーム
powder
光学活性
[α]/D -16 to -22°, c = 0.5 in methanol
色
white to beige
溶解性
DMSO: 2 mg/mL, clear
保管温度
2-8°C
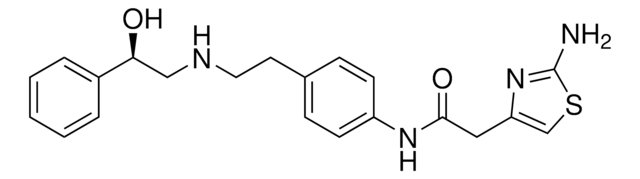
SMILES記法
O[C@@H](CNCCC1=CC=C(NC(CC2=CSC(N)=N2)=O)C=C1)C3=CC=CC=C3
InChI
1S/C21H24N4O2S/c22-21-25-18(14-28-21)12-20(27)24-17-8-6-15(7-9-17)10-11-23-13-19(26)16-4-2-1-3-5-16/h1-9,14,19,23,26H,10-13H2,(H2,22,25)(H,24,27)/t19-/m0/s1
InChI Key
PBAPPPCECJKMCM-IBGZPJMESA-N
類似した製品をお探しですか? 訪問 製品比較ガイド
アプリケーション
Mirabegron has been used as a β3-adrenergic receptor (β3-AR) agonist in an in vitro experiment to determine whether β3-AR stimulation specifically activates brown adipose tissue (BAT) thermogenesis in humans.
生物化学的/生理学的作用
Mirabegron is a non-antimuscarinic orally active drug with the potential to treat patients with overactive bladder (OAB) symptoms. It aids in bladder relaxation by acting on neural control of the storage phase of micturition by targeting β3-AR. Mirabegron is also used to treat neurogenic detrusor overactivity (NDO) in pediatric patients.
Mirabegron is a potent and selective β3-adrenoceptor agonist that activates the β3 adrenergic receptor in the detrusor muscle in the bladder leading to muscle relaxation and an increase in bladder capacity.
シグナルワード
Warning
危険有害性情報
危険有害性の分類
Repr. 2
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
SML2480-VAR:
SML2480-5MG:
SML2480-BULK:
SML2480-25MG:
最新バージョンのいずれかを選択してください:
試験成績書(COA)
Lot/Batch Number
この製品を見ている人はこちらもチェック
Toshiyuki Takasu et al.
The Journal of pharmacology and experimental therapeutics, 321(2), 642-647 (2007-02-13)
We evaluated the pharmacological characteristics of (R)-2-(2-aminothiazol-4-yl)-4'-{2-[(2-hydroxy-2-phenylethyl)amino]-ethyl} acetanilide (YM178). YM178 increased cyclic AMP accumulation in Chinese hamster ovary (CHO) cells expressing human beta3-adrenoceptor (AR). The half-maximal effective concentration (EC50) value was 22.4 nM. EC50 values of YM178 for human beta1-
Rebecca Bragg et al.
The Consultant pharmacist : the journal of the American Society of Consultant Pharmacists, 29(12), 823-837 (2014-12-19)
To review the literature regarding the efficacy and safety of mirabegron for the treatment of overactive bladder (OAB). A literature search was performed using MEDLINE (PubMed) prior to December 31, 2013, using the terms "mirabegron" and "randomized-controlled trial." All published
Pradeep Tyagi et al.
Expert opinion on drug safety, 10(2), 287-294 (2010-12-15)
Mirabegron is being developed as a new treatment for the management of overactive bladder (OAB). It is an orally active drug that works by activating the β(3)-adrenoceptor with a better safety profile than antimuscarinic drugs. However, long-term adverse effects are
Kentaro Konishi et al.
European journal of drug metabolism and pharmacokinetics, 43(3), 301-309 (2017-11-23)
Mirabegron is cleared by multiple mechanisms, including drug-metabolizing enzymes. One of the most important clearance pathways is direct glucuronidation. In humans, M11 (O-glucuronide), M13 (carbamoyl-glucuronide), and M14 (N-glucuronide) have been identified, of which M11 is one of the major metabolites
Susan J Keam
Paediatric drugs, 23(4), 411-415 (2021-06-01)
Mirabegron (MYRBETRIQ®), a beta-3 adrenergic agonist developed by Astellas Pharma Inc., is well established as a treatment for overactive bladder in adults and is available as extended-release (ER) tablets administered once daily. More recently, mirabegron has been investigated in pediatric
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)