PHR1573
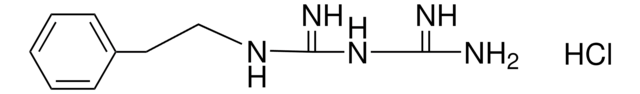
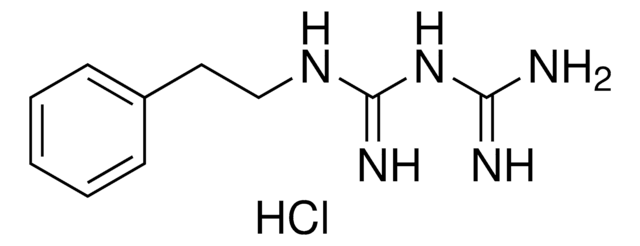
Phenformin Hydrochloride
Pharmaceutical Secondary Standard; Certified Reference Material
동의어(들):
Phenformin hydrochloride, N-(2-Phenylethyl)imidodicarbonimidic diamide monohydrochloride, Phenethylbiguanide
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C10H15N5 · HCl
CAS Number:
Molecular Weight:
241.72
EC Number:
MDL number:
UNSPSC 코드:
41116107
PubChem Substance ID:
NACRES:
NA.24
추천 제품
Grade
certified reference material
pharmaceutical secondary standard
Quality Level
API family
phenformin
CofA
current certificate can be downloaded
포장
pkg of 500 mg
기술
HPLC: suitable
gas chromatography (GC): suitable
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-30°C
SMILES string
Cl.NC(=N)NC(=N)NCCc1ccccc1
InChI
1S/C10H15N5.ClH/c11-9(12)15-10(13)14-7-6-8-4-2-1-3-5-8;/h1-5H,6-7H2,(H6,11,12,13,14,15);1H
InChI key
YSUCWSWKRIOILX-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards. Phenformin Hydrochloride is the hydrochloride salt of phenformin, which belongs to the second chemical class of oral hypoglycemic agents such as biguanides.
애플리케이션
Phenformin Hydrochloride may be used as a pharmaceutical reference standard for the quantification of the analyte in pharmaceutical formulations using surface enhanced Raman spectroscopy technique and spectrophotometric technique.
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
분석 메모
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
기타 정보
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
각주
To see an example of a Certificate of Analysis for this material enter LRAB7788 in the slot below. This is an example certificate only and may not be the lot that you receive.
추천 제품
Find a digital Reference Material for this product available on our online platform ChemisTwin® for NMR. You can use this digital equivalent on ChemisTwin® for your sample identity confirmation and compound quantification (with digital external standard). An NMR spectrum of this substance can be viewed and an online comparison against your sample can be performed with a few mouseclicks. Learn more here and start your free trial.
이미 열람한 고객
Principles of Endocrine Pharmacology, 59(1), 18-18 (2012)
Direct spectrophotometric analysis of glipizide and phenformin hydrochloride in pharmaceutical dosage forms
Phalke.NP, et al.
Indian Journal of Pharmaceutical Sciences, 59(1), 18-18 (1997)
Analysis of drugs illegally added into Chinese traditional patent medicine using surface-enhanced Raman scattering
Zhang Y, et al.
Analytical Sciences, 29(10), 985-990 (2013)
Seon-Hyeong Lee et al.
Cancers, 12(6) (2020-06-03)
Lung adenocarcinoma cells express high levels of ALDH1L1, an enzyme of the one-carbon pathway that catalyzes the conversion of 10-formyltetrahydrofolate into tetrahydrofolate and NAD(P)H. In this study, we evaluated the potential of ALDH1L1 as a therapeutic target by deleting the
Benjamin D Stein et al.
Journal of proteome research, 18(10), 3703-3714 (2019-08-10)
Recent advances in genome editing technologies have enabled the insertion of epitope tags at endogenous loci with relative efficiency. We describe an approach for investigation of protein interaction dynamics of the AMP-activated kinase complex AMPK using a catalytic subunit AMPKα2
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.