280909
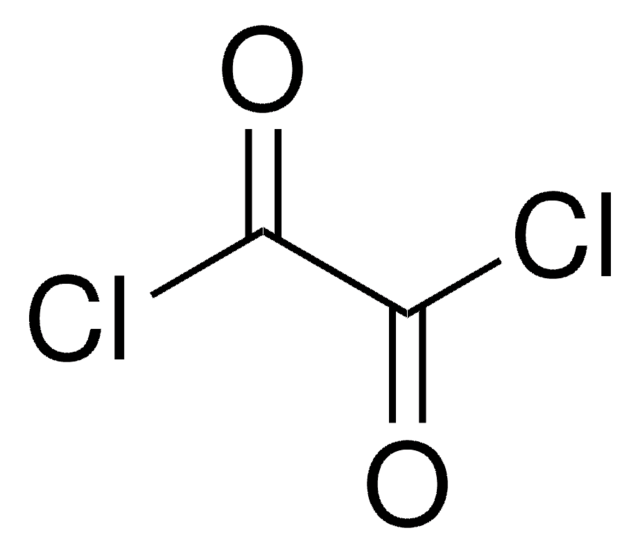
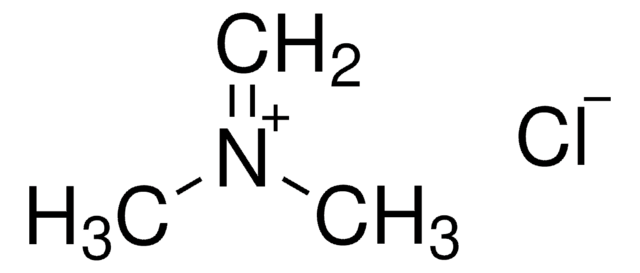
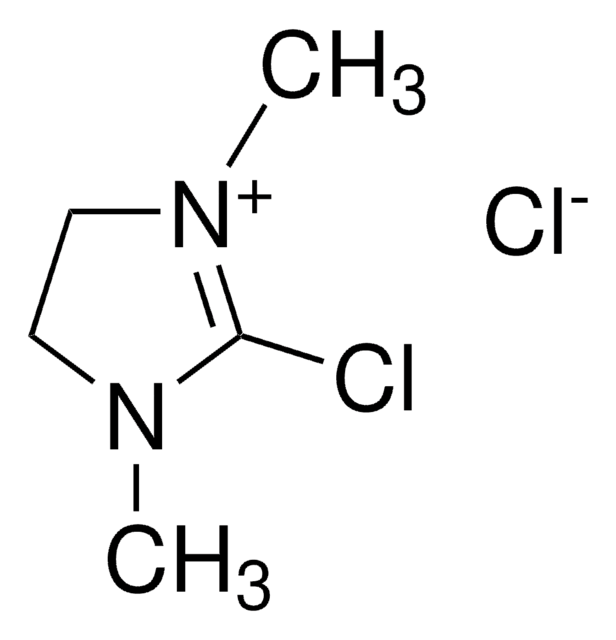
(Chloromethylene)dimethyliminium chloride
95%
Synonym(s):
(Chlormethylene)dimethylammonium chloride, Arnold′s reagent, Dimethylchloroformiminium chloride, Vilsmeier reagent
About This Item
Recommended Products
assay
95%
form
solid
reaction suitability
reaction type: C-C Bond Formation
impurities
<5% DMF
mp
132 °C (dec.) (lit.)
functional group
amine
chloro
storage temp.
2-8°C
SMILES string
[Cl-].C\[N+](C)=C\Cl
InChI
1S/C3H7ClN.ClH/c1-5(2)3-4;/h3H,1-2H3;1H/q+1;/p-1
InChI key
QQVDYSUDFZZPSU-UHFFFAOYSA-M
Looking for similar products? Visit Product Comparison Guide
Application
- β-Lactams vis the Staudinger reaction
- N-vinyl substituted indoles via cross coupling of NH-indoles with vinyl bromides
- Multi ring-fused 2-pyridone-based fluorescent scaffold
- ester prodrugs as anti-poxvirus agents
- Trimethine cyanine dyes for sensing G-quadruplex formation
- Phospha-isosteres via Hundsdiecker-Barton iododecacarboxylation
- 2-Azetidinones via [2+2]-cycloaddition
Used for studying organic photosensitizers for application in dye-sensitized solar cells
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Repr. 1B - Skin Corr. 1A
supp_hazards
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service