W296902
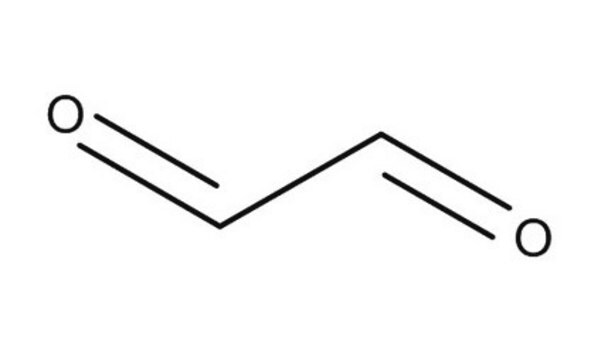
Pyruvaldehyde solution
40 wt. % in H2O
Synonym(s):
Methylglyoxal solution, Acetylformaldehyde, Pyruvaldehyde, Pyruvic aldehyde
About This Item
Recommended Products
biological source
synthetic
Quality Level
grade
Halal
concentration
40 wt. % in H2O
refractive index
n20/D 1.41
density
1.178 g/mL at 25 °C
application(s)
flavors and fragrances
documentation
see Safety & Documentation for available documents
food allergen
no known allergens
organoleptic
caramel
storage temp.
2-8°C
SMILES string
[H]C(=O)C(C)=O
InChI
1S/C3H4O2/c1-3(5)2-4/h2H,1H3
InChI key
AIJULSRZWUXGPQ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Disclaimer
signalword
Danger
hcodes
Hazard Classifications
Eye Dam. 1 - Met. Corr. 1 - Muta. 2 - Skin Sens. 1
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service