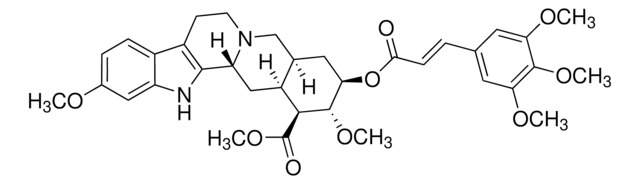
おすすめの製品
形状
powder
品質水準
色
white to light brown
溶解性
DMSO: 10 mg/mL, clear
SMILES記法
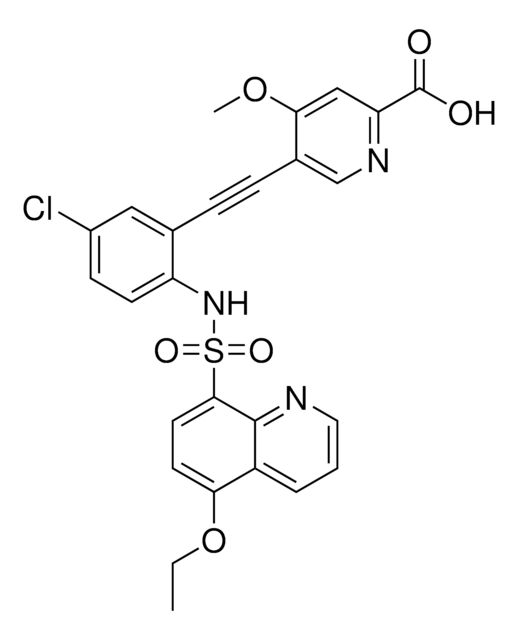
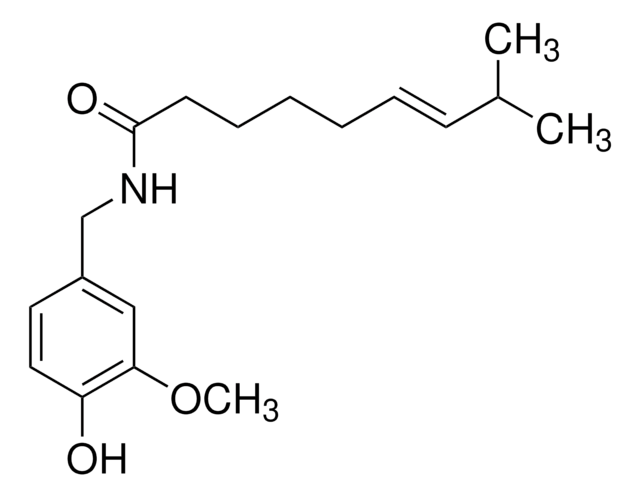
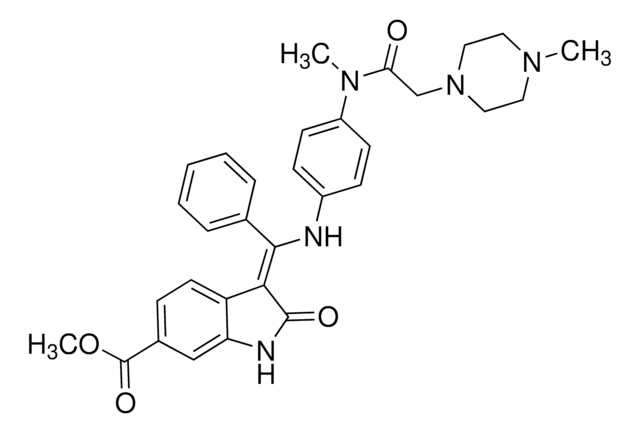
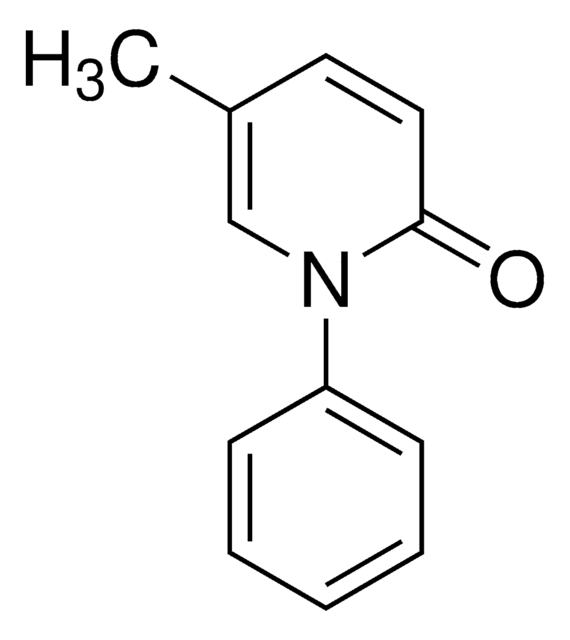
O=C(OC)[C@@H]1[C@]2([H])C[C@@]3([H])C4=C(C5=CC=C(OC)C=C5N4)CCN3C[C@]2([H])C[C@H](OC(C6=CC(OC)=C(OC(OCC)=O)C(OC)=C6)=O)[C@H]1OC
InChI
1S/C35H42N2O11/c1-7-46-35(40)48-31-26(42-3)12-18(13-27(31)43-4)33(38)47-28-14-19-17-37-11-10-22-21-9-8-20(41-2)15-24(21)36-30(22)25(37)16-23(19)29(32(28)44-5)34(39)45-6/h8-9,12-13,15,19,23,25,28-29,32,36H,7,10-11,14,16-17H2,1-6H3/t19-,23+,25-,28-,29+,32+/m1/s1
InChI Key
ZCDNRPPFBQDQHR-SSYATKPKSA-N
アプリケーション
- to study its effects on anti-CD147-induced metabolon disruption in human breast cancer cells
- to study its effects on Cryptosporidium parvum-infected HCT-8 cells
- in orthogonal linear separation analysis (OLSA)-derived decomposed analysis
生物化学的/生理学的作用
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
SML1908-BULK:
SML1908-25MG:
SML1908-5MG:
SML1908-VAR:
試験成績書(COA)
製品のロット番号・バッチ番号を入力して、試験成績書(COA) を検索できます。ロット番号・バッチ番号は、製品ラベルに「Lot」または「Batch」に続いて記載されています。
この製品を見ている人はこちらもチェック
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)